PINNA (AURICLE)
The pinna is composed of fibroelastic cartilage covered by skin. This cartilage is continuous with that of the external auditory canal (EAC). Anteriorly the cartilage is adherent to the skin, but posteriorly there is a loose areolar layer in between. The dimensions of characteristics of the pinna include (Fig. 1.1)1,2 a height of 5–6 cm, width 55% of height, forms an angle that is 20° from the vertical plane, has an auricle that diverges from the occipital scalp at 21°–30°, forms an angle of < 90° at the conchal bowl to the mastoid and has a distance of 1–2 cm from the helical rim to the mastoid.
There are two intrinsic and three extrinsic ligaments. There are six intrinsic and six extrinsic muscles. The major intrinsic ligaments attach from the crus helicis to the tragus and from the antihelix to the cauda helicis. The extrinsic ligaments are named anterior (from the tragus to the crus of helicis to the zygoma), superior (from the spine of the helix to the superior EAC), posterior medial (from the concha to the mastoid prominence). The muscles are divided into intrinsic (helix major, helix minor, tragus, antitragus, transverse, oblique) and extrinsic (auricularis posterior, auricularis superior, auricularis anterior).
The pinna and mastoid scalp area are supplied by branches of the external carotid artery (Fig. 1.2). Veins also accompany the named arteries. The postauricular artery3 supplies the posterior surface of the pinna, except the lobule and the mastoid scalp. It has auricular, mastoid, transverse nuchal artery4 branches. The occipital artery5 supplies part of the mastoid scalp (via a lateral branch) and has a medial and a lateral branch. The superficial temporal artery6 supplies the anterior pinna, the temporal scalp, and the superficial temporalis fascia. Its branches are7 anterior (or superior), the temporal artery (via the main trunk or parietal branch), parietal, temporal, and middle temporal (Fig. 1.3).
The pinna is formed from the sixth gestational week from the six hillocks of His that originate from the first and second branchial arches (Fig. 1.1). It is completely formed by week 16. It reaches adult size by age five. Hillocks one to three are supplied by the auriculotemporal nerve and hillocks four to six are supplied by a branch from facial nerve, greater auricular and lesser occipital nerves (Figs. 1.4A and B).8,92
The external ear is important for sound localization.10 The folds and crevices that create the shape of the pinna facilitate sound localization in vertical space. This design provides a 10 kHz dip with elevation of sound. In contrast, sound localization in horizontal space is provided by three mechanisms. The resonance gain of the near ear provides the gain of the ear closest to the sound. The head shadow effect attenuates frequencies <2 kHz so that there is a 5–15 dB interaural difference. There is an interaural time difference of 0.6 ms between sounds that is detected within the ascending brainstem nuclei.
EXTERNAL AUDITORY CANAL
The EAC is composed of cartilage in its lateral third (8 mm) and bone in its medial two thirds (16 mm). The cartilaginous EAC is deficient superiorly but there is a ligament between the helix and tragus. The EAC runs inferiorly and anteriorly in adults but more horizontally in neonates. The anterior wall of the EAC is longer than the posterior wall by 4 mm. It contains two sutures: the tympanosquamous (anteriorly) and the tympanomastoid (posteriorly) with the skin in the intervening area forming the “vascular strip”. The EAC contains two constrictions, at the bony cartilaginous junction and at the isthmus. The isthmus is 5 mm lateral to the tympanic membrane (TM) produced from the anterior canal overhang. The tympanic ring is deficient superiorly at the tympanic incisura.11
Figs. 1.4A and B: The embryology of the pinna. The six hillocks of His form as follows: one (tragus), two (helix crus), three (helix), four (antihelix), five (antitragus), and six (lobule, inferior helix).
The EAC is largely supplied by the deep auricular artery, a branch from the internal maxillary artery that passes through the parotid, running posterior to temporomandibular joint (TMJ). The “vascular strip” is supplied by the anterior/superior auricular artery, a branch from the superficial temporal artery. The skin of the EAC is much thinner in the bony canal and lacks appendages (cerumen, sweat glands, and hair follicles). The cartilaginous EAC skin contains these appendages with cerumen glands being most prominent in the superior and inferior walls. The EAC is drained by the superficial temporal vein and postauricular vein.11 The lymph drains mainly to parotid nodes and mastoid nodes.
The EAC is formed by ectoderm of the first pouch as it contacts the endoderm of first pouch with intervening mesoderm that forms the TM. The bony EAC is formed by four centers of ossification of the tympanic ring in the ninth week.
TYMPANIC MEMBRANE
The TM is a trilaminar structure composed of an outer epidermal layer, middle connective tissue layer, and an inner mucosal layer. It forms an angle with the superior EAC of 140° and the inferior EAC of 55°. Its dimensions are 8 mm (horizontal) by 10 mm (vertical). It is composed of a pars tensa and a pars flaccida that has a thicker connective tissue layer. The epithelium of the TM migrates radially from the umbo at 1–2 mm per day.13 Upon perforation, keratinocytes form from progenitor cells at the umbo and migrate across to close the perforation.14,15 The middle connective tissue layer contains collagen fibers arranged in three patterns: radial, circular, and oblique (Fig. 1.5).
The lateral surface of the TM is supplied by the deep auricular artery. The medial surface is supplied by the anterior tympanic artery, stylomastoid artery (a branch from the postauricular artery) and also by branches of the middle meningeal artery.16 The lateral surface of the TM is supplied by the auriculotemporal nerve from cranial nerve (CN) V, auricular branch of CN X (Arnold's nerve), and branches from CN VII, IX, X. The medial surface is supplied by CN VII and CN IX branches from the tympanic plexus.
The TM is formed by ectoderm (epithelial layer) and endoderm (mucosal layer) with intervening mesoderm between these two layers (connective tissue layer) of the first branchial pouch.
MIDDLE EAR
The middle ear is divided into areas above the level of the superior TM annulus (epitympanum), between the superior and inferior TM annulus (mesotympanum) and below the inferior TM annulus (hypotympanum). The protympanum refers to the area anterior to a vertical line level with the anterior margin of the annulus. The posterior tympanum is the area posterior to a vertical line at the posterior margin of the annulus. It includes the facial recess and sinus tympani. The middle ear contains three mucocilliary tracts that all drain toward the Eustachian tube. These are the hypotympanic (the largest tract), the epitympanic, and the promontorial tracts.
The walls of the middle ear contain some important landmarks. Its lateral wall is composed of the tympanic bone and TM. It has three holes. The posterior canaliculus for chorda18 lies between the junction of posterior and lateral wall at the level of the malleus handle. The Petrotympanic (Glaserian) fissure19 transmits the anterior malleolar ligament, the anterior tympanic artery, and the chorda tympani nerve (via the canal of Huguier). The roof of the middle ear is composed of the Petrous and Squamous bones. It has one hole, the petrosquamous suture19 that transmits veins to the inferior petrosal sinus. The anterior wall has three holes. These communicate the superior and inferior caroticotympanic nerves (sympathetic branches from the carotid to the tympanic plexus) and the tympanic branches of the internal carotid artery. There are also two tunnels, the canal for the tensor tympani muscle and the Eustachian tube. On the medial wall is the oval window (fenestra vestibulae) and round window (fenestra cochleae). The oval window dimensions are 3.25 mm by 1.75 mm and contain the stapes footplate that has dimensions of 3 mm by 1.4 mm. The round window is closed by the secondary TM that communicates to the floor of the scala tympani of the cochlea. The round window membrane20,21 has dimensions of 2.3 mm by 1.87 mm. It is composed of three layers: mucosal, fibrous, and an inner mesothelium. It is directed inferior and posterior. It sits within a niche22 that is triangular in shape with anterior (1.5 mm), posterior superior (1.3 mm), and posterior inferior (1.6 mm) walls. Also on the middle wall is the facial nerve. The facial nerve runs in the middle ear from the first turn at the geniculate ganglion to the tympanic segment to the second genu and then to the mastoid (vertical) segment. The facial nerve may be dehiscent (defined as a nonpathological gap ≥ 0.4 mm) in the middle ear with an incidence 55% There is an incidence of more than one dehiscence in 22%. Dehiscences occur 90% at the tympanic segment (with 80% just above oval window and 25% also involving facial nerve prolapse) It may also occur next to tensor tendon, in the facial recess or in the anterior epitympanic recess. The remainder of the dehiscences (10%) occurs in the mastoid segment (Fig. 1.6).23–27
The posterior wall28 of the middle ear contains the fossa incudis that itself contains the short process of the incus and a ligament connecting to the incus. The facial recess lies between the pyramidal process, facial nerve, and the TM. The sinus tympani29 is bounded medially by the posterior semicircular canal and laterally by the pyramidal eminence and the facial nerve. It is bounded superiorly by the ponticulus (a ridge running from the pyramidal eminence to the oval window) and the lateral semicircular canal and inferiorly by the subiculum, styloid eminence, and the jugular wall (Fig. 1.7).
Fig. 1.6: The lateral wall of the middle ear and the path of the tympanic facial nerve. (RW, round window; HSCC, horizontal semicircular canal; PSCC, posterior semicircular canal; GSPN, greater superficial petrosal nerve).From C Gralapp and R Jackler, Stanford University, CA, USA.
The posterior wall of the mesotympanum also contains three eminences and three ridges (Fig. 1.8).
The epitympanum contains the head of the malleus, body of the incus, the malleolar and incudal ligaments and folds. The epitympanum communicates with the mastoid air cells via the mastoid antrum. The mastoid antrum is fully developed at birth with a volume of 1 mL. The supratubal recess (or anterior tympanic recess) is bounded by30,31: anteriorly, the petrosal tegmen, posteriorly by the cog (a ridge of bone extending down from the tegmen), superiorly by the middle cranial fossa tegmen, and laterally by the tympanic bone and chorda tympani nerve.
The malleus32 consists of a head, neck, lateral and anterior process and manubrium. The lateral process attaches to the TM and the anterior process provides attachment for the anterior malleolar ligament to the petrotympanic fissure. The manubrium provides attachment of the malleus to the TM from the lateral process to the umbo. It has three ligaments. The anterior malleolar attaches the head to the anterior wall of epitympanum, the lateral malleolar attaches the neck to the notch of Rivinus, and the superior malleolar attaches the head to the roof of the epitympanum.6
The malleus also has three associated folds; an anterior malleolar fold attaching the lateral process to the annulus, a posterior malleolar fold attaching the lateral process to the annulus, and a lateral malleolar fold attaching to the annulus (Figs. 1.9A to D).
The tensor tympani has an origin with its muscle fibers within the canal for the tensor tympani, turning to tendon that loops 90° around the cochleariform process33 to insert onto the neck of malleus. It is innervated by the nerve to tensor tympani (a branch from CN V).
The incus34 is composed of a body, short process, long process, and a lenticular process that articulates with stapes. The ligaments of the incus are the posterior incudal that attaches the short process to the incudal recess (in the area of the “incus buttress”) posteriorly, a medial and lateral that attaches the body to the head of the malleus, and a superior incudal ligament which is variably present.
The stapes35 parts are the head (capitulum) for articulation with the lenticular process of the incus and an anterior and posterior crus. The anterior crus is straighter, shorter, and thinner. The stapes footplate has dimensions of 1.75 mm by 3.25 mm. Its relations to the structures within the vestibule are important to consider and differ depending on the area of the footplate. The utricle is at 0.58 mm (posterior), 1.04 mm (middle) and 1.51 mm (anterior), compared to the saccule which is at 1.33 mm (middle) and 1.31 mm (anterior) (Fig. 1.10).36
The stapedius muscle33 originates from the pyramidal process and inserts into the posterior crus of stapes just below its head. It is innervated by the nerve to stapedius (from CN VII). It has a number of hypothesized functions.37,38 The stapedius protects from loud sounds by contracting at sounds louder than 80 dBHL with a latency of >10 ms. It is most efficient at 2 kHz. It provides rigidity and blood supply to the ossicular chain and reduces physiological noise from chewing and talking. The stapedial reflex39 is the contraction of the stapedial muscle that occurs bilaterally to sound presented to one ear. The stapedial reflex is a measure of the integrity of the inner hair cells (IHCs). Contraction of the stapedial muscle occurs at approximately 70–100 (mean 85) dBHL above threshold. The ipsilateral reflex occurs at approximately 2–14 dB less than the contralateral ear. Stimulating both ears lowers the threshold by 3 dB. If a patient has hearing loss combined with the reflex threshold greater than the limits of the audiometer (approximately 120 dB), the reflex will be absent. It is also absent in 5–20% of normal people (Fig. 1.11).
Fig. 1.10: The relationships of the stapes footplate to the utricle and the saccule.Source: Adapted from Backous et al.36
The tympanic diaphragm is a series of mucosal folds that separate the epitympanum from the mesotympanum and the mastoid. Its components are the malleus head, body of incus, lateral incudal fold, medial incudal fold, anterior malleolar fold, lateral malleolar fold, and tensor tympani fold.40 There are two narrow passages that breach the diaphragm, the anterior and posterior tympanic isthmuses. The anterior tympanic isthmus is medial to the body of the incus, passing between the stapes, long process incus, and the tensor tympani tendon. The posterior tympanic isthmus is between the pyramidal process, short process of incus and posterior incudal ligament, medial incudal fold, stapes and stapedial tendon (Fig. 1.12).
Fig. 1.12: The tympanic diaphragm and connections between the epitympanun and the mesotympanum. Areas of communication are highlighted.Source: Adapted from Proctor.40
In surgery for chronic otitis media, sometimes the head of the malleus and the body of the incus are removed with the attaching folds and ligaments to increase aeration between the middle ear and mastoid.
Beneath the floor of the attic and in the upper mesotympanum there are three compartments.40 The inferior incudal space is from the inferior surface of the incus laterally to the posterior malleolar fold limited medially by the medial incudal fold and anteriorly by the interossicular fold. The posterior pouch of von Troeltsch is between the TM and the posterior malleolar fold. Its inferior edge often contains the chorda tympani. It opens inferiorly toward the posterior mesotympanum. The anterior pouch of von Troeltsch is between the TM and the anterior malleolar fold.
The chorda tympani18,26 runs in the mesotympanum and supplies taste to the anterior two-thirds of the tongue and is secretomotor to the submandibular and sublingual glands. The taste cell bodies lie in the geniculate ganglion. It leaves the facial nerve, on average, 5 mm (a range of −1 to 11 mm) above the stylomastoid foramen and runs in the canaliculus of chorda tympani. It enters the middle ear via the ita chordae posterius (in the posterior lateral wall of the middle ear) with the posterior tympanic artery. It lies between the pyramidal eminence and the tympanic annulus. It runs through the middle ear lateral to the long process of the incus and the tendon of tensor tympani but medial to the neck of the malleus and posterior malleolar ligament. It exits the middle ear at the iter chordae anterius to run in the canal of Huguier. The chorda tympani may have a number of variations. In the middle ear it may pass lateral to the TM or may pass lateral to the malleus. In the mastoid it may arise distal to the stylomastoid foramen or may arise as high as the lateral semicircular canal.
The tympanic plexus42 lies on the cochlea promontory. The incoming fibers include parasympathetics that come from Jacobson's nerve (CN IX) and Arnold's nerve (CN X) and sympathetic branches from the internal carotid artery Its outgoing fibers include the lesser superficial petrosal nerve that carry parasympathetic branches of CN IX. It leaves in a small canal beneath the tensor tympani. It is joined by parasympathetic branches of CN VII and emerges lateral to the greater superficial petrosal nerve in the middle fossa before leaving by the foramen ovale. The plexus also has a branch to the greater superficial petrosal nerve and TM mucosal branches.
The hypotympanum lies beneath the floor of the bony EAC and contains hypotympanic air cells with the jugular bulb posterior inferiorly and the internal carotid artery anterior inferiorly. Either may be dehiscent in the hypotympanum.
The blood supply of the middle ear45 is by branches of the external carotid, internal carotid, and basilar arteries. The branches of the external carotid artery branches include the anterior tympanic artery that runs from the internal maxillary artery through the petrotympanic fissure. It has a superior branch that supplies the anterior lateral epitympanum, a posterior branch that supplies the TM, incudostapedial joint and lateral tegmen, and an ossicular branch. The ossicular branch further divides into a mallear and incudal branch. The external carotid also gives the deep auricular artery via the maxillary internal maxillary artery through the inferior bony EAC. It has an anterior and posterior branch. The inferior tympanic artery runs from the ascending pharyngeal artery through the inferior tympanic canaliculus. The stylomastoid artery runs 9from the postauricular artery running up through the stylomastoid foramen. The stylomastoid artery also provides the posterior tympanic artery. There is a mastoid branch from the occipital artery. A tubal branch comes from the accessory meningeal artery. The superficial petrosal artery branches from the middle meningeal artery and runs with the greater superficial petrosal nerve. A superior tympanic artery branches from the middle meningeal artery through the superior tympanic canaliculus with the lesser superficial petrosal nerve. The internal carotid artery supplies blood to the middle ear via its caroticotympanic branches2 that come direct from the internal carotid artery. The basilar artery supplies the middle ear via branches of the subarcuate artery, itself coming from the labyrinthine artery, or the anterior inferior cerebellar artery.
The inferior tympanic artery is the usual blood supply to a glomus tympanicum tumor.47
The middle ear and Eustachian tube forms from the expansion of the first pouch. The epitympanum and antrum are developed by birth. The expansion of the first pouch leads to the development of four primary sacs.40 The saccus medius forms the epitympanum and divides into three smaller spaces, the medial (to later form Prussak's space), the posterior, and the anterior. The saccus anticus forms the anterior pouch of von Troeltsch. The saccus superior forms the posterior pouch of von Troeltsch. The saccus posticus forms the posterior middle ear and hypotympanum (Fig. 1.13).
The resonant frequencies of the external and middle ears are determined by their design.50 This results in a combined external ear gain of 15–20 dB over 2–7 kHz and 5 dB at 3 kHz in the middle ear. The resonance of the middle ear is includes the air space contained within the mastoid antrum. This effect is lost by blocking the antrum, as is sometimes performed in chronic ear surgery. The combined resonance of the external and middle ear is thought to produce the typical notch in the audiogram seen in sensory hearing loss associated with noise induced hearing loss. The gain provided via the various parts of the ear include51 the concha with a 10 dB gain at 5 kHz and the EAC with a 10 dB gain at 2–5 kHz. In infants the EAC provides a 15 dB gain at 3 kHz (at approximately 8 kHz in infants) and reaches adult value after age two and a half. The middle ear52 has a gain of 5 dB at 3 kHz and the TM has a gain at 800–1600 Hz.
Sound is transferred from the TM to the cochlear with a gain according to the transformer ratio theory.53,54 It accounts for a total gain of approximately 25–30 dB. In normal subjects up to 25 dB variations in middle ear mechanics can occur. At 1 kHz, the ratio of gain from the TM to the cochlear is 82.5. This is a combination of three levers: the hydraulic, ossicular, and catenary. The hydraulic lever provides a gain of 17–20 times relates to the ratio of the area of the oval window to the TM. The ossicular lever has a gain of 1.3 represents the ratio of the long axis of the malleus to the long process of the incus. The catenary lever provides a gain of 2 due to the outward convexity of TM with its radial arrangement of collagen fibers.
The levers have greatest efficiency at 1 kHz. Frequencies > 1 kHz lead to a reduction in the efficiency of the levers. Above 1 kHz, the hydraulic lever changes as vibrations are broken up into smaller vibratory patterns, while the ossicular lever provides more slippage in the axis of rotation of the ossicles. The TM has its greatest movement at its inferior edge at 2 kHz. Above 6 kHz vibrations are broken up into small zones. The annular ligament provides approximately 90% of the stiffness. No movement occurs at the malleoincudal joint at physiological sound pressures.
INNER EAR
The cochlea is 5 mm in height. The cochlea duct is 34 mm and has two and three quarter turns around the helicotrema. The modiolus points laterally, anteriorly, and inferiorly. It has a similar orientation as the EAC.56 The cochlea aqueduct transmits the periotic duct (perilymphatic duct) between the basal turn of the scala tympani to the media of the jugular fossa (Table 1.1 and Fig. 1.14).21
Hair cells62,63 do not possess true stereocilia, which require a nine plus two arrangement of microtubules and do not have kinocilia. They are like microvilli with an actin core. They increase in length along the cochlea duct. The first-order neurons of the cochlea nerve lie in the spiral ligament, compared to within the IAC for the vestibular nerve. The IHCs are arranged to form a single row of hair cells and are flask shaped. They have only afferent synapses. Their stereocilia do not attach into the tectorial membrane, they are deflected by fluid in between tectorial membrane and reticular lamina. Cilia deflection causes mechanically gated ion channels to open allowing potassium and calcium to enter the cell. This then causes voltage gated calcium channels to open, causing depolarization. Outer hair cells (OHCs)64,65 are arranged in three rows and are cylinder shaped. They have both afferent and efferent synapses. They are unique to mammals. Their stereocilia attach into the bottom surface of the tectorial membrane. They are responsible for the sensitivity of hearing and provide the “cochlea amplifier”. They do this by sensing the incoming sound wave, and then generating force in synchrony with it to increase the vibration of the basilar membrane.11
This provides a gain of up to 60 dB. The plasma membrane of the OHC contains prestin, which is a motor protein that senses the voltage within the cell and generates force. This causes the length of the OHC to elongate and contract, a phenomenon called electromotility. By adding this additional energy to the cochlear traveling wave, viscous damping forces within the cochlea are overcome, leading to high-frequency hearing.
The basilar membrane has a tonotopic organization. This is primarily because its stiffness decreases from base to apex, although its width (and hence its mass) also increases along the length of the cochlea. The traveling wave of the basilar membrane vibrates with maximum amplitude at a place along the cochlea that is dependent on the frequency of the sound presented. The corresponding hair cells stimulate the adjacent nerve fibers, which are organized according to the frequency at which they are most sensitive.12
The lower frequencies travel further (to the apex). The basilar membrane response allows complex sounds to be broken up into narrow bands of frequencies.
The cochlea fluids67,68 are predominantly composed of potassium (K+) and sodium (Na+). The difference in potassium concentrations between the compartments leads to an endocochlear potential of +80–100 mV. This is maintained by a Na+/K+ ATPase within the stria vascularis (Table 1.2).
The arterial supply45,69,70 (Fig. 1.15) of the inner ear is via branches of the labyrinthine artery (a branch of the anterior inferior cerebellar artery, but sometimes comes from the basilar or superior cerebellar artery). It runs in the IAC before becoming the anterior vestibular artery. This will supply the utricle, superior semicircular canal, and lateral semicircular canal (the embryological pars superior)). From this, the common cochlear artery branches. This branches again into two further arteries, the main cochlear artery and the vestibulocochlear artery. The main cochlear artery supplies the apical three quarters of the cochlea and the modiolus. It gives the external radiating arterioles, which gives four networks supplying the cochlea. The spiral ligament branches give branches to the scala vestibule and the scala tympani. The other networks are to the stria vascularis and the spiral prominence. The main cochlea artery also gives the internal radiating arterioles that provide the limbus vessels and marginal vessels. The vestibulocochlear artery gives the posterior vestibular artery that supplies the saccule and posterior semicircular canal (the embryological pars inferior). It also gives the cochlear ramus artery to supply the basal one quarter of the cochlea.
| |||||||||||||||||||||
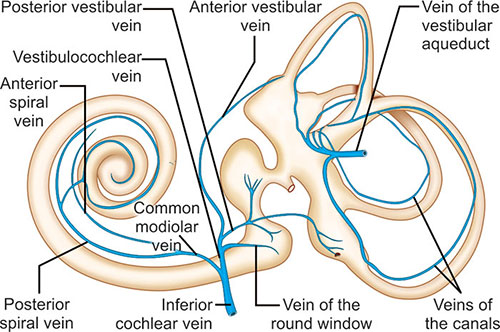
The venous drainage of the inner ear (Fig. 1.16) is similarly matched to the arterial supply. The anterior spiral vein, draining the spiral ligament and scala vestibule, and the posterior spiral vein, draining the scala media and scala tympani, both drain into the common modiolar vein. The anterior vestibular vein, draining the pars superior, the posterior vestibular vein, draining the pars inferior, and the vein of the round window all drain into the vestibulocochlear vein. The common modiolar vein joins the vestibulocochlear vein to form the inferior cochlear vein, which then drains to the inferior petrosal vein via the canal of Cotugno.13
Fig. 1.16: The arterial supply of the cochlea.72
The membranous semicircular canals drain into the vein of the vestibular aqueduct that then joins the sigmoid sinus. Sometimes there is also an internal auditory vein that flows into the inferior petrosal sinus via the IAC (Fig. 1.17).
Cochlear neurons can be divided into type I or type II.72 Afferent nerve fibers are unmyelinated and run from the organ of Corti, through the habenula perforata, to the spiral ganglion. Spiral ganglion neurons are bipolar and acquire the myelin in the modiolus. This can be compared to the vestibular neurons that are myelinated and gain their myelin sheath as they cross the basement membrane directly under the sensory epithelium. 95% of the afferent nerve fibers synapse with IHCs. These are called type I afferent neurons, and are the primary source of auditory input to the brain. Type II afferent neurons receive input from OHCs, and their purpose is unknown. Efferent fibers are the terminations of descending olivocochlear nerve fibers (Rasmussen's bundle). They come from the brainstem and synapse onto the OHCs. Their function is unknown at this time, but they may be important to understanding speech in noisy background environments. The sensory epithelium in the inner can be divided according to embryological origin into the pars superior (supplying the superior semicircular canal, lateral semicircular canal and utricle) and pars inferior (supplying the posterior semicircular canal and saccule) (Table 1.3).
The endolymphatic fluid73 is produced predominantly by the stria vascularis but also by the planum semilunatum (around cristae), the dark vestibular cells and from the perilymph across the labyrinth membranes. Vasopressin plays a role in its formation. It moves by longitudinal flow and radial exchange. Longitudinal flow is slow at 0.004–0.007 mm/min and flows from the stria vascularis to the scala media to the ductus reunions to the saccule and them to the endolymphatic sac. Radial exchange is rapid and occurs via an exchange and balance of chemicals.
Fig. 1.17: The venous drainage of the cochlea.72
| |||||||||||||||||||||||||
INTERNAL AUDITORY CANAL
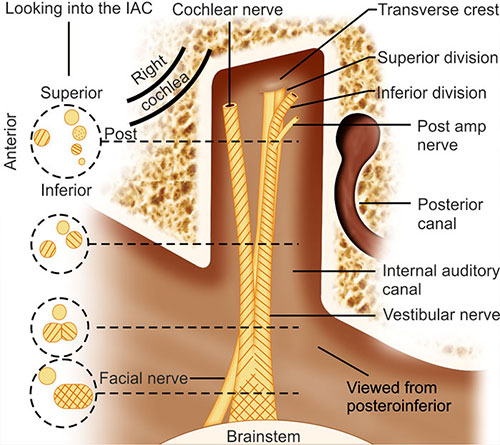
The IAC has a diameter of 4.5 mm with a <1 mm difference between sides. If a CT scan demonstrates that there is a >2 mm difference in an individual between sides, it is suggestive of a pathological condition (such as a vestibular schwannoma). Its length along the posterior wall measures 8 mm with <2 mm between sides. It runs at an angle of 80°–90° (in 60%) or 90°–100° (in 40%) to the sagittal plane.74 Anatomical variations include an absence of the bony partition on its lateral end associated with a stapes gusher. It may be too narrow (<3 mm). If this occurs with normal facial nerve function it typically means there is coexisting cochlea nerve aplasia. It may have a vertical orientation, associated with hearing loss. The IAC may also 14be too wide (>10 mm) and associated with a stapes gusher. The IAC contains the facial nerve, cochlear nerve, superior vestibular nerve, inferior vestibular nerve, internal auditory artery (labyrinthine artery), the singular nerve (branches from the inferior vestibular nerve at the fundus and passes through its own foramen to supply the posterior semicircular canal), the intracanalicular cistern and meninges.75 The relations of the contents of the IAC are important.76 At the brainstem the cochlear nerve is below all, with the facial nerve anterior and the vestibular nerve posterior. At the porus,77 the facial nerve is above all, the vestibular nerve is posterior inferior and the cochlear nerve is anterior superior. The relationship at the fundus is shown in Figure 1.18. The nerves rotate 90° within the canal and the cerebellopontine angle so that the facial nerve is superior to the vestibulocochlear nerves and is separated at the brainstem by the anterior inferior cerebellar artery.76
There are at least two acousticofacial anastomoses within the IAC between the nervus intermedius and the superior vestibular nerve (Figs. 1.19 and 1.20).78
Fig. 1.18: The relationships of the nerves within the internal auditory canal.Source: Adapted from Rhoton and Tedeschi.77
Embryologically, the vestibular system develops before the cochlea.79 The inner ear develops from the otic placode that forms on each side of the neural groove between the second and third branchial arches. The otic placode then forms the otic pit and then the otic vesicle. This then develops into two compartments. The medial compartment will form the endolymphatic sac. The lateral compartment will form the utriculosaccular chamber, which, after undergoing a series of complex folds, forms two chambers: the utricular and the saccular chamber. The utricular chamber forms the utricle and semicircular canals. The superior forms first, then the posterior with the lateral canal forming last. As the lateral semicircular canal develops the slowest, it is the most commonly maldeveloped canal. The saccular chamber forms the saccule and cochlear.
This explains the connection between them called the ductus reunions. The cochlea develops by intrachondral bone within endochondral bone and therefore heals by fibrous healing. At gestational week 20 the cochlea reaches adult length. At week 25 the organ of Corti is fully developed. The bony labyrinth develops as mesoderm and surrounds the membranous labyrinth. Ossification then occurs from 14 ossification centers from week 15. The vestibular aqueduct develops from a diverticulum in the otocyst in week 4 (Table 1.4 and Fig. 1.21).
Inner ear malformations81–83 can be classified as membranous/osseous or membranous. Membranous malformations account for 80% of congenital deafness. Membranous/osseous malformations can be further classified as total aplasia (Michel's) or partial. Cochlear deformities can be complete aplasia, hypoplasia, common cavity (the most common), or Mondini's malformations. Mondini's malformation is described as a cochlea with only one and a half turns, a large vestibule, normal labyrinth, and enlarged vestibular aqueduct (EVAS). It is the most common malformation and is associated with other syndromes including Pendred's, branchial-oto-renal, Waardenburg's, Wildervancks (cervico-oculo-acoustic syndrome), Klippel Feil (fusion of cervical vertebrae), and Treacher Collins. Lateral semicircular canal dysplasia is the most common labyrinthine abnormality as it is the last canal to develop. The most common radiological abnormality is EVAS. The cochlear aqueduct is thought to be abnormal when >1 mm. It is usually patent, but neither transmits pressure or allows free spinal fluid to flow. Membranous abnormalities can either be incomplete or complete. Scheibe is an incomplete type, cochleosaccular dysplasia (of the pars inferior) with a malformed membranous canal. The organ of Corti is partially or totally missing with a normal bony canal. Alexander is also an incomplete type and is partial (localized to the basal turn) aplasia of cochlea duct occurring with high-frequency sensory hearing loss. Bing Siebemann malformations involve complete membranous dysplasia and are associated with Jervell and Lange-Nielsen syndrome and Usher's syndrome (Table 1.5 and Fig. 1.22).
|
|
AUDITORY CORTEX
The auditory cortex is in the lateral (Sylvian) fissure.84 It is arranged with low frequencies superior and high frequencies inferior. The auditory cortex exhibits plasticity in that it has the ability to modify or reorganize. Although this plasticity occurs throughout life, it occurs to a greater extent at younger ages. Modification of the tonotopic organization of the auditory cortex occurs after hearing loss and with rehabilitation of hearing (Fig. 1.23).
REFERENCES
- Petersson RS, Friedman O. Current trends in otoplasty. Curr Opin Otolaryngol Head Neck Surg. 2008;16(4):352–8.
- Adamson PA, Litner JA. Otoplasty technique. Otolaryngol Clin North Am. 2007;40(2):305–18.
- Imanishi N, Nakajima H, Aiso S. Arterial anatomy of the ear. Okajimas Folia Anat Jpn. 1997;73(6):313–23.
- McKinnon BJ, Wall MP, Karakla DW. The vascular anatomy and angiosome of the posterior auricular artery. A cadaver study. Arch Facial Plast Surg. 1999;1(2):101–4.
- Alvernia JE, Fraser K, Lanzino G. The occipital artery: a microanatomical study. Neurosurgery. 2006;58(1 suppl): ONS114–22; discussion ONS-22.
- Song R, Song Y, Qi K, et al. The superior auricular artery and retroauricular arterial island flaps. Plast Reconstr Surg. 1996;98(4):657–67; discussion 68–70.
- Wright CG. Development of the human external ear. J Am Acad Audiol. 1997;8(6):379–82.
- Klockars T, Rautio J. Embryology and epidemiology of microtia. Facial Plast Surg. 2009;25(3):145–8. Epub 2009/ 10/08.
- Whitfield IC. Mechanisms of sound localization. Nature. 1971;233(5315):95–7. Epub 1971/09/10.
- * Kelly KE, Mohs DC. The external auditory canal. Anatomy and physiology. Otolaryngol Clin North Am. 1996;29(5): 725–39.
- Chandler JR. Malignant external otitis and osteomyelitis of the base of the skull. Am J Otol. 1989;10(2):108–10.
- Alberti PW. Epithelial migration on the tympanic membrane. J Laryngol Otol. 1964;78:808–30.
- Santa Maria PL, Redmond SL, McInnes RL, et al. Tympanic membrane wound healing in rats assessed by transcriptome profiling. Laryngoscope. 2011;121(10):2199–213. doi: 10.1002/lary.22150.
- Santa Maria PL, Redmond SL, Atlas MD, et al. Histology of the healing tympanic membrane following perforation in rats. Laryngoscope. 2010;120(10):2061–70.
- Applebaum EL, Deutsch EC. Fluorescein angiography of the tympanic membrane. Laryngoscope. 1985;95(9 Pt 1): 1054–8.
- Mehta RP, Rosowski JJ, Voss SE, et al. Determinants of hearing loss in perforations of the tympanic membrane. Otol Neurotol. 2006;27(2):136–43.
- Gray O. The chorda tympani. J Laryngol Otol. 1953;67(3): 128–38.
- Proctor B, Nielsen E, Proctor C. Petrosquamosal suture and lamina. Otolaryngol Head Neck Surg. 1981;89(3 Pt 1): 482–95.
- Bellucci RJ, Fisher EG, Rhodin J. Ultrastructure of the round window membrane. Laryngoscope. 1972;82(6):1021–6.
- Su WY, Marion MS, Hinojosa R, et al. Anatomical measurements of the cochlear aqueduct, round window membrane, round window niche, and facial recess. Laryngoscope. 1982;92(5):483–6.
- Stewart TJ, Belal A. Surgical anatomy and pathology of the round window. Clin Otolaryngol Allied Sci. 1981;6(1): 45–62.
- Monkhouse WS. The anatomy of the facial nerve. Ear Nose Throat J. 1990;69(10):677–83, 86–7.
- Proctor B, Nager GT. The facial canal: normal anatomy, variations and anomalies. I. Normal anatomy of the facial canal. Ann Otol Rhinol Laryngol Suppl. 1982;97:33–44.
- Sando I, English GM, Hemenway WG. Congenital anomalies of the facial nerve and stapes: a human temporal bone report. Laryngoscope. 1968;78(3):316–23.
- Nager GT, Proctor B. The facial canal: normal anatomy, variations and anomalies. II. Anatomical variations and anomalies involving the facial canal. Ann Otol Rhinol Laryngol Suppl. 1982;97:45–61.
- * Proctor B. The anatomy of the facial nerve. Otolaryngol Clin North Am. 1991;24(3):479–504.
- Proctor B. Surgical anatomy of the posterior tympanum. Ann Otol Rhinol Laryngol. 1969;78(5):1026–40.
- Donaldson JA, Anson BJ, Warpeha RL, et al. The surgical anatomy of the sinus tympani. Arch Otolaryngol. 1970;91 (3):219–27.
- Petrus LV, Lo WW. The anterior epitympanic recess: CT anatomy and pathology. AJNR Am J Neuroradiol. 1997;18 (6):1109–14.
- Gluth MB, Cohen MA, Friedland PL, et al. Surgical anatomy of the anterior supralabyrinthine air cell tract. J Laryngol Otol. 2011;125(10):1009–13. Epub 2011/06/15.
- Richany SF, Anson BJ, Bast TH. The development and adult structure of the malleus, incus and stapes. Q Bull Northwest Univ Med Sch. 1954;28(1):17–45.
- Anson BJ, Donaldson JA, Warpeha RL, et al. The surgical anatomy of the ossicular muscles and the facial nerve. Laryngoscope. 1967;77(8):1269–94.
- Todd NW, Creighton FX Jr. Malleus and incus: correlates of size. Ann Otol Rhinol Laryngol. 2013;122(1):60–5.
- Anson BJ, Bast TH. Anatomical structure of the stapes and the relation of the stapedial footplate to vital parts of the otic labyrinth. Ann Otol Rhinol Laryngol. 1958;67(2):389–99.
- Backous DD, Minor LB, Aboujaoude ES, et al. Relationship of the utriculus and sacculus to the stapes footplate: anatomic implications for sound-and/or pressure-induced otolith activation. Ann Otol Rhinol Laryngol. 1999;108(6):548–53.
- Margolis RH, Levine SC. Acoustic reflex measures in audiologic evaluation. Otolaryngol Clin North Am. 1991;24(2): 329–47.
- Borg E, Counter SA. The middle-ear muscles. Sci Am. 1989; 261(2):74–80.
- Hall CM. Stapedial reflex decay in retrocochlear and cochlear lesions. Review of procedures and methods for conducting SRD tests. Ann Otol Rhinol Laryngol. 1977;86 (2 pt. 1):219–22.
- * Proctor B. The development of the middle ear spaces and their surgical significance. J Laryngol Otol. 1964;78:631–48.
- McMinn RMH, Last RJ. Last's anatomy: regional and applied, 9th edn. Elsevier Health Sciences; Edinburgh: 1994. 705p.
- Tekdemir I, Aslan A, Tuccar E, et al. An anatomical study of the tympanic branch of the glossopharyngeal nerve (nerve of Jacobson). Ann Anat. 1998;180(4):349–52.
- Daud AS, Pahor AL. Tympanic neurectomy in the management of parotid sialectasis. J Laryngol Otol. 1995;109(12): 1155–8.
- Thomas RL. Tympanic neurectomy and chorda tympani section. Aust N Z J Surg. 1980;50(4):352–5. Epub 1980/08/01.
- Janfaza P, Nadol JB Jr, Galla RJ, et al. Surgical anatomy of the head and neck. Cambridge, MA: Harvard University Press; 2011. 932 p.
- Pahor AL, Hussain SS. Persistent stapedial artery. J Laryngol Otol. 1992;106(3):254–7. Epub 1992/03/01.
- Hesselink JR, Davis KR, Taveras JM. Selective arteriography of glomus tympanicum and jugulare tumors: techniques, normal and pathologic arterial anatomy. AJNR Am J Neuroradiol. 1981;2(4):289–97.
- * Jackler RK. The surgical anatomy of cholesteatoma. Otolaryngol Clin North Am. 1989;22(5):883–96.
- Palva T, Northrop C, Ramsay H. Aeration and drainage pathways of Prussak's space. Int J Pediatr Otorhinolaryngol. 2001;57(1):55–65. Epub 2001/02/13.
- Huttenbrink KB. The mechanics of the middle-ear at static air pressures: the role of the ossicular joints, the function of the middle-ear muscles and the behaviour of stapedial prostheses. Acta Otolaryngol Suppl. 1988;451:1–35.
- Caiazzo AJ, Tonndorf J. Ear canal resonance and temporary threshold shift. Otolaryngology. 1978;86(5):ORL–820.
- Garcia-Gonzalez A, Gonzalez-Herrera A. Effect of the middle ear cavity on the response of the human auditory system. J Acoust Soc Am. 2013;133(5):3544.
- Koike T, Wada H, Kobayashi T. Modeling of the human middle ear using the finite-element method. J Acoust Soc Am. 2002;111(3):1306–17.
- Gan RZ, Reeves BP, Wang X. Modeling of sound transmission from ear canal to cochlea. Ann Biomed Eng. 2007;35 (12):2180–95.
- Sichel JY, Freeman S, Sohmer H. Lateralization during the Weber test: animal experiments. Laryngoscope. 2002; 112(3):542–6.
- Toth M, Alpar A, Bodon G, et al. Surgical anatomy of the cochlea for cochlear implantation. Ann Anat. 2006;188(4): 363–70.
- Gopen Q, Rosowski JJ, Merchant SN. Anatomy of the normal human cochlear aqueduct with functional implications. Hear Res. 1997;107(1–2):9–22.
- Kimura RS. The ultrastructure of the organ of Corti. Int Rev Cytol. 1975;42:173–222.
- Lim DJ. Cochlear anatomy related to cochlear micromechanics: A review. J Acoust Soc Am. 1980;67(5):1686–95.
- * Lim DJ. Functional structure of the organ of Corti: a review. Hear Res. 1986;22:117–46.
- Harada Y, Tagashira N, Takunida M, et al. Three-dimensional ultrastructure of cochlea: a review. Adv Otorhinolaryngol. 1990;45:49–68.
- Roberts WM, Howard J, Hudspeth AJ. Hair cells: transduction, tuning, and transmission in the inner ear. Ann Rev Cell Biol. 1988;4:63–92.
- Meyer AC, Moser T. Structure and function of cochlear afferent innervation. Curr Opin Otolaryngol Head Neck Surg. 2010;18(5):441–6.
- Brownell WE. Outer hair cell electromotility and otoacoustic emissions. Ear Hear. 1990;11(2):82–92.
- Dallos P. The role of outer hair cells in cochlear function. Prog Clin Biol Res. 1985;176:207–30.
- Shulman A, Goldstein B, Strashun AM. Final common pathway for tinnitus: theoretical and clinical implications of neuroanatomical substrates. Int Tinnitus J. 2009;15(1): 5–50. Epub 2009/10/22.
- Zorowka PG. Otoacoustic emissions: a new method to diagnose hearing impairment in children. Eur J Pediatr. 1993;152(8):626–34.
- Guinan JJ, Jr, Salt A, Cheatham MA. Progress in cochlear physiology after Bekesy. Hear Res. 2012;293(1–2):12–20.
- Schratzenstaller B, Janssen T, Alexiou C, et al. Confirmation of G. von Bekesy's theory of paradoxical wave propagation along the cochlear partition by means of bone-conducted auditory brainstem responses. ORL J Otorhinolaryngol Relat Spec. 2000;62(1):1–8.
- Anniko M, Wroblewski R. Ionic environment of cochlear hair cells. Hear Res. 1986;22:279–93.
- Waltner JG, Raymond S. On the chemical composition of the human perilymph and endolymph. Laryngoscope. 1950;60(9):912–8.
- Anson BJ, Winch TR, Warpeha RL, et al. The blood supply of the otic capsule of the human ear, with special reference to that of the cochlea. Ann Otol Rhinol Laryngol. 1966;75(4): 921–44.
- Anson BJ, Warpeha RL, Rensink MJ. The gross and macroscopic anatomy of the labyrinths. Ann Otol Rhinol Laryngol. 1968;77(4):583–607.
- Mazzoni A. The vascular anatomy of the vestibular labyrinth in man. Acta Otolaryngol Suppl. 1990;472:1–83.
- Spoendlin H. Anatomy of cochlear innervation. Am J Otolaryngol. 1985;6(6):453–67.
- Wangemann P. K+ cycling and the endocochlear potential. Hear Res. 2002;165(1–2):1–9. Epub 2002/05/29.
- Rhoton AL, Jr, Tedeschi H. Microsurgical anatomy of acoustic neuroma. Otolaryngol Clin North Am. 1992;25(2): 257–94.
- Mazzoni A, Hansen CC. Surgical anatomy of the arteries of the internal auditory canal. Arch Otolaryngol. 1970;91(2): 128–35.
- Silverstein H, Norrell H, Haberkamp T, et al. The unrecognized rotation of the vestibular and cochlear nerves from the labyrinth to the brain stem: its implications to surgery of the eighth cranial nerve. Otolaryngol Head Neck Surg. 1986;95(5):543–9.
- Mazzoni A. Internal auditory canal arterial relations at the porus acusticus. Ann Otol Rhinol Laryngol. 1969;78(4): 797–814.
- Unel S, Yilmaz M, Albayram S, et al. Anastomoses of the vestibular, cochlear, and facial nerves. J Craniofac Surg. 2012;23(5):1358–61.
- Pujol R, Lavigne-Rebillard M, Uziel A. Development of the human cochlea. Acta Otolaryngol Suppl. 1991;482:7–12; discussion 3.
- Freeman S, Geal-Dor M, Sohmer H. Development of inner ear (cochlear and vestibular) function in the fetus-neonate. J Basic Clin Physiol Pharmacol. 1999;10(3):173–89.
- * Jackler RK, Luxford WM, House WF. Congenital malformations of the inner ear: a classification based on embryogenesis. Laryngoscope. 1987;97(3 Pt 2 Suppl 40):2–14.
- Suehiro S, Sando I. Congenital anomalies of the inner ear: introducing a new classification of labyrinthine anomalies. Ann Otol Rhinol Laryngol Suppl. 1979;88(4 Pt 3 Suppl 59): 1–24.
- Sennaroglu L, Saatci I. A new classification for cochleovestibular malformations. Laryngoscope. 2002;112(12):2230–41.
- Nieuwenhuys R. Anatomy of the auditory pathways, with emphasis on the brain stem. Adv Otorhinolaryngol. 1984; 34:25–38.
- Borg E. Physiological mechanisms in auditory brainstem-evoked response. Scand Audiol Suppl. 1981;13:11–22.
- Miller CA, Brown CJ, Abbas PJ, et al. The clinical application of potentials evoked from the peripheral auditory system. Hear Res. 2008;242(1–2):184–97.
- Luxon LM. The anatomy and pathology of the central auditory pathways. Br J Audiol. 1981;15(1):31–40.
- *References with asterisks refer to critical sources.