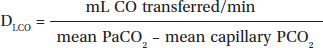ANATOMY
Geetanjali S Verma
The important anatomy to the airway consists of the relationship of pharynx to its surrounding structures, larynx and mobility of tissues.
THE PHARYNX
Extension: Sphenoid bone to C6
12 to 15 cm long
It is widest at the level of the hyoid bone (5 cm) and narrowest at the level of the esophagus (1.5 cm), which is the most common site for obstruction after foreign body aspiration.
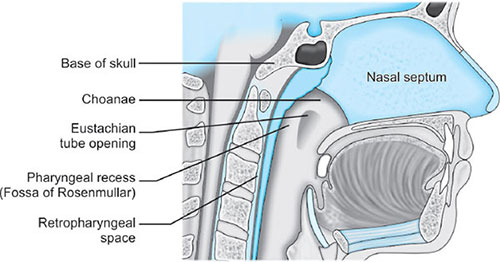
Lies parallel to vertebra, covered by anterior/longitudinal ligament and fascial layers beneath mucosa and constrictor muscles. The retropharyngeal space (between superficial buccopharyngeal fascia and prevertebral fascia) permits free movement of pharynx during deglutition. Retropharngeal abscesses may infiltrate to superior mediastinum through here.
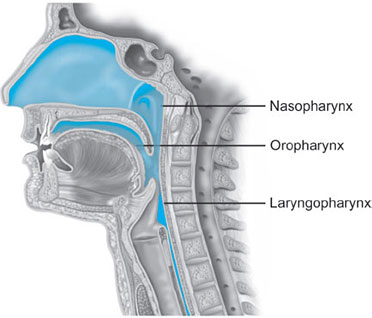
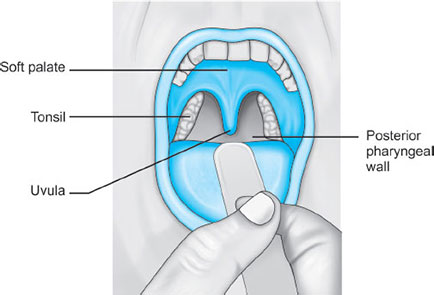
Parts (Fig. 1)
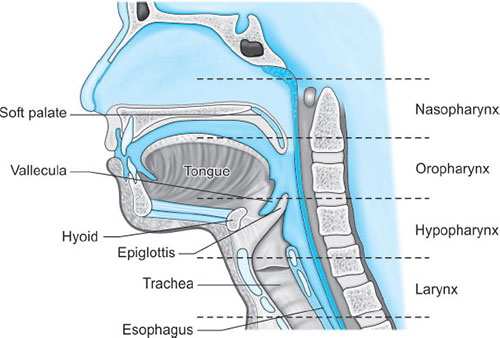
- Nasopharynx: Extends from skull base to soft palate at caudal aspect of C1
- Oropharynx: Extension of nasopharynx to caudal aspect of C3; also involves anterior 1/3 to posterior 2/3rd of tongue
- Layngopharynx (hypopharynx): Merges with esophagus at C6, where the cricopharyngeus encircles the esophagus to form its upper sphincter (similar function of Sellick's maneuver in anesthetized patients).
Nasopharynx
The nose: The nose is divided into two nasal fossae which extend uo to 10 to 14 cm from the nostrils to the nasopharynx. The two fossae are divided by a cartilaginous septum. The nasal septum is composed of the perpendicular plate of the ethmoid bone descending from the cribriform plate, septal cartilage, and the vomer.
Disruption of the cribriform plate (due to facial trauma/head injury) may allow direct communication with the anterior fossa. Use of positive-pressure mask ventilation in such conditions may lead to the entry of bacteria or foreign material, causing meningitis or sepsis. Also there is a probability of nasal airways, nasotracheal tubes, and nasogastric tubes being introduced into the subarachnoid space.
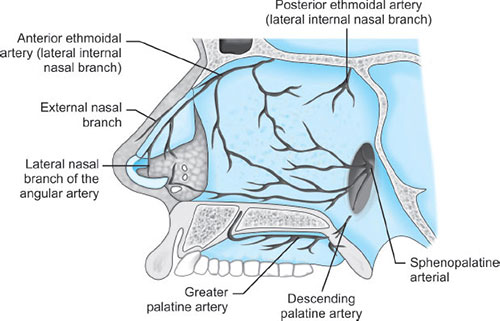
Blood Supply (Fig. 3)
- Ethmoid branches of the ophthalmic artery
- Sphenopalatine and greater palatine branches of the maxillary artery
- Superior labial and lateral nasal branches of the facial artery.
Kiesselbach's plexus, where these vessels anastomose, is situated in Little's area on the anterior-inferior portion of the nasal septum—a source of significant epistaxis.
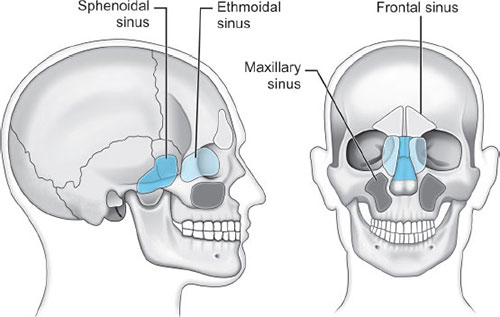
Paranasal sinuses: Sphenoid, ethmoid, maxillary, and frontal (Fig. 4).
Prolonged nasotracheal intubation has most often been associated with infection of the maxillary sinus as its drainage is hindered by the location of the ostia superiorly in the sinus promoting a chronic infectious process.
Nerve supply (Fig. 5): The olfactory area consists of the middle and upper septum and the superior turbinate bone, located in the upper third of the nasal fossa. The olfactory cells have specialized hairlike processes (olfactory hair) innervated by the olfactory nerve.
The respiratory portion is located in the lower third of the nasal fossa.
Trigeminal nerve (1st 2 divisions) supplies the nonolfactory sensory area.
The parasympathetic autonomic nerves reach the mucosa from the facial nerve after relaying through the sphenopalatine ganglion, and sympathetic fibers are derived from the plexus surrounding the internal carotid artery through the vidian nerve.
Oropharynx
It starts below the soft palate, and extends to the superior edge of the epiglottis (Fig. 6).
Anterior wall is formed by the posterior third of the tongue.
During anesthesia or sedation with the patient in supine position, muscle relaxation + gravity = movement of base of the tongue toward the posterior oropharyngeal wall, causing airway obstruction. This is managed by use of oral airways or jaw lift.
The oropharynx opens to the oral cavity at the palatoglossal folds, marking the division between the anterior two-thirds and posterior one-third of the tongue. The palatoglossal folds make the fauces, which contain the tonsils.5
Hypertrophied tonsils can cause a challenge during mask ventilation or intubation.
Anterior to the fauces is the oral cavity proper, separated from the vestibule by the teeth and gums. Prominent or bucked maxillary teeth can interfere with laryngoscopy and intubation.
Laryngopharynx
Lies opposite C3-6 vertebrae
Consists of 3 paired and 3 unpaired cartilages, supporting muscles and membranes (Fig. 7).
Paired: Arytenoids, corniculate, cuneiform.
Unpaired: Epiglottis, thyroid, cricoid.
Adult male larynx | Adult female larynx | |
|---|---|---|
Length | 44 | 36 |
Transverse diameter | 43 | 41 |
Sagittal diameter | 36 | 26 |
CARTILAGES
Hyoid bone: U shaped, 2.5 cm wide, 1 cm thick, has greater and lesser horns (cornu).
It is attached to the styloid processes of the temporal bones by the stylohyoid ligament and to the thyroid cartilage by the thyrohyoid membrane and muscle. Intrinsic tongue muscles originate on the hyoid, and the pharyngeal constrictors are attached here.6
Thyroid Cartilage
Named so for its shield-like shape (from embryologic midline fusion of the two distinct quadrilateral laminae). In females, the sides join at approximately 120 degrees, and in males is approx 90 degrees (Adam's apple). The thyroid notch lies in the midline at the top of the fusion site of the two laminae. On the inner side of this fusion line are attached the vestibular ligaments and, below them, the vocal ligaments. The superior (greater) and inferior (lesser) cornu of the thyroid are the slender posteriorly directed extensions of the edges of the lamina. The lateral thyrohyoid ligament attaches the superior cornu to the hyoid bone, and the cricoid cartilage articulates with the inferior cornu at the cricothyroid joint. The movements of this joint are rotatory and gliding, which leads to changes in the length of the vocal folds.
Cricoid Cartilage
The tracheal rings connect to the cricoid by ligaments and muscles and it attaches to the thyroid cartilage by the cricothyroid membrane (a site for percutaneous or sugical cricothyroidotomy).
The superior thyroid artery, the superior and inferior thyroid veins and the jugular veins were reported to traverse the membrane.
Arytenoid Cartilage
These are shaped like three-sided pyramids, and they lie in the posterior aspect of the larynx. The base of the arytenoid is concave and articulates by a true diarthrodial joint with the superior lateral aspect of the posterior lamina of the cricoid cartilage. The lateral extension of the arytenoid base is called the muscular process, where intrinsic laryngeal muscles, lateral and posterior cricoarytenoids originate. The medial extension of the arytenoid 7base is called the vocal process. Vocal ligaments, the bases of the true vocal folds, extend from the vocal process to the midline of the inner surface of the thyroid lamina. Broyles’ ligament connects the vocal ligament to the thyroid cartilage and contains lymphatics and blood vessels (acts as a source for extension of laryngeal cancer outside the larynx).
Epiglottis
It is shaped like a leaf and is found between the larynx and the base of the tongue.
The upper border of the epiglottis is attached by its narrow tip to the midline of the thyroid cartilage by the thyroepiglottic ligament. The hyoepiglottic ligament connects the epiglottis to the back of the body of the hyoid bone. The mucous membrane that covers the anterior aspect of the epiglottis sweeps forward to the tongue as the median glossoepiglottic fold and to the pharynx as the paired lateral pharyngoepiglottic folds. The pouch-like areas found between the median and lateral folds are the valleculae (site of impaction of foreign body)
Cuneiform and Corniculate Cartilages
The epiglottis is connected to the arytenoid cartilages by the laterally placed aryepiglottic ligaments and folds. Two sets of paired fibroelastic cartilages are embedded in each aryepiglottic fold. The sesamoid cuneiform cartilage is roughly cylindrical and lies anterosuperior to the corniculate in the fold. The cuneiform may be seen laryngoscopically as a whitish elevation through the mucosa. The cuneiform and corniculate cartilages reinforce and support the aryepiglottic folds and may help the arytenoids move.
Laryngeal Cavity
Extends from the laryngeal inlet to the lower border of the cricoid cartilage. It consists of the superiorly placed vestibular folds (false cords), and vocal folds (true vocal cords). The space between the true cords is called the rima glottidis, or the glottis. The glottis is divided into two parts—anterior intermembranous section (situated between the two vocal folds) and posterior intercartilaginous part (which passes between the two arytenoid cartilages and the mucosa, stretching between them in the midline posteriorly, forming the posterior commissure of the larynx).
The area extending from the laryngeal inlet to the vestibular folds is known as the vestibule or supraglottic larynx. The laryngeal space from the free border of the cords to the cricoid cartilage is called the subglottic or infraglottic larynx.
The region between the vestibular folds and the glottis is termed the ventricle or the sinus. The ventricle may expand anterolaterally to a pouch-like area with many lubricating mucous glands called the laryngeal saccule. The pyriform sinus lies laterally to the aryepiglottic fold within the inner surface of the thyroid cartilage.8
MUSCLES (FIG. 8)
Extrinsic Muscles of the Larynx
Muscle | Function | Innervation |
|---|---|---|
Sternohyoid | Indirect depressor of the larynx | Cervical plexus |
Ansa hypoglossi C1, C2, C3 | ||
Sternothyroid | Depresses the larynx Modifies the thyrohyoid and aryepiglottic folds | Same as above |
Thyrohyoid | Same as above | Cervical plexus Hypoglossal nerve C1, C2 |
Thyroepiglottic | Mucosal inversion of aryepiglottic fold | Recurrent laryngeal nerve |
Stylopharyngeus | Assists folding of thyroid cartilage | Glossopharyngeal |
Inferior pharyngeal constrictor | Assists in swallowing | Vagus, pharyngeal plexus |
Source: Benumof JL: Airway Management—Principles and Practice. | ||
Intrinsic Musculature of the Larynx (Fig. 9)
Muscle | Function | Innervation |
|---|---|---|
Posterior cricoarytenoid | Abductor of vocal cords | Recurrent laryngeal |
Lateral cricoarytenoid | Adducts arytenoids closing glottis | Recurrent laryngeal |
Transverse arytenoid | Adducts arytenoids | Recurrent laryngeal |
Oblique arytenoid | Closes glottis | Recurrent laryngeal |
Aryepiglottic | Closes glottis | Recurrent laryngeal |
Vocalis | Relaxes the cords | Recurrent laryngeal |
Thyroarytenoid | Relaxes tension cords | Recurrent laryngeal |
Cricothyroid | Tensor of the cords | Superior laryngeal (external branch) |
Source: Benumof JL: Airway Management—Principles and Practice. | ||
BLOOD SUPPLY (FIG. 10)
The external carotid gives rise to the superior thyroid artery, which bifurcates, forming the superior laryngeal artery. This artery courses with the superior laryngeal nerve through the thyrohyoid membrane to supply the supraglottic region.
The inferior thyroid artery (from thyrocervical trunk), terminates as the inferior laryngeal artery. This vessel travels in the tracheoesophageal groove with the recurrent laryngeal nerve and supplies the infraglottic larynx. There are extensive connections with the ipsilateral superior laryngeal artery and across the midline. A small cricothyroid artery may branch from the superior thyroid and cross the cricothyroid membrane. It most commonly travels near the inferior border of the thyroid cartilage.10
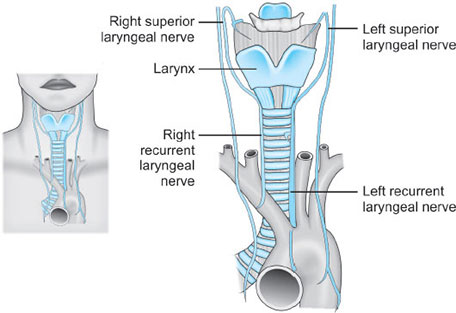
NERVES (FIG. 11)
The main nerves of the larynx are the recurrent laryngeal nerves and the internal and external branches of the superior laryngeal nerves (from 11vagus nerve). The external branch of the superior laryngeal nerve supplies motor innervation to the cricothyroid muscle. All other motor supply to the laryngeal musculature is provided by the recurrent laryngeal nerve.
The recurrent laryngeal nerve also provides sensory innervation to the larynx below the vocal cords.
VOCAL CORD PALSY
There is an intimate and important relationship between the nerves that supply the larynx and the vessels that supply the thyroid gland.
The external branch of the superior laryngeal nerve descends over the inferior constrictor muscle of the pharynx immediately deep to the superior thyroid artery and vein as these pass to the superior pole of the gland; at this site the nerve may be damaged in securing these vessels (Figs. 12A and B).
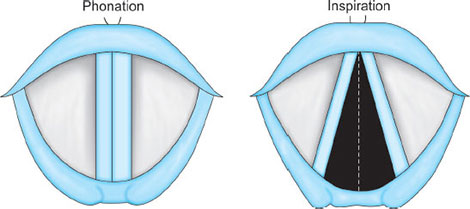
The glottic chink appears oblique during phonation. The aryepiglottic fold on the affected side appears shortened and the one on the normal side is lengthened. The cords may appear wavy. The symptoms include frequent throat clearing and difficulty in raising the vocal pitch. A total bilateral paralysis of vagus nerves affects the recurrent laryngeal nerves and the superior laryngeal nerves. In this condition, the cords assume the abducted, cadaveric position. The vocal cords are relaxed and appear wavy. A similar picture may be seen following the use of muscle relaxants.
The recurrent laryngeal nerve, as it ascends in the tracheoesophageal groove, is overlapped by the lateral lobe of the thyroid gland, and here comes into close relationship with the inferior thyroid artery as this passes medially, behind the common carotid artery, to the gland. The artery may cross posteriorly or anteriorly to the nerve, or the nerve may pass between the terminal branches of the artery. On the right side, there is an equal chance of locating the nerve in each of these three situations; on the left, the nerve is more likely to lie posterior to the artery.
Fig. 12A: Position of vocal cords during phonation and inspiration.Source: Hodder Headline PLC, London.
Fig. 12B: Diagrammatic representation of different types of vocal cord palsies. Note that in complete bilateral recurrent laryngeal palsy (bottom), vocal cords remain in the abducted position and the glottic opening is preserved.Source: Hodder Headline PLC, London.
Injury to the recurrent nerve is an obvious hazard of thyroidectomy, especially since the nerve may be displaced from its normal anatomical location by a diseased thyroid gland. Recurrent laryngeal nerve paralysis may occur not only as a result of injury at thyroidectomy but also from involvement of the nerve by a malignant or occasionally benign enlargement of the thyroid gland, by enlarged lymph nodes or by cervical trauma. The recurrent laryngeal nerve carries both abductor and adductor fibers to the vocal cords. The abductor fibers are more vulnerable, and moderate trauma causes a pure abductor paralysis (Selmon's law). Severe trauma causes both abductor and adductor fibers to be affected. Pure adductor paralysis does not occur as a clinical entity. In the case of pure unilateral abductor palsy, both cords meet in the midline on phonation (since adduction is still possible on the affected side). However, only the normal cord abducts during inspiration.13
In the case of complete unilateral palsy of the recurrent laryngeal nerve, both abductors and adductors are affected. On phonation, the unaffected cord crosses the midline to meet its paralyzed counterpart, appearing to lie in front of the affected cord. On inspiration, the unaffected cord moves to full abduction. When abductor fibers are damaged bilaterally (incomplete bilateral damage to the recurrent laryngeal nerve), the adductor fibers draw the cords toward each other and the glottic opening is reduced to a slit, resulting in severe respiratory distress. However, with a complete palsy, each vocal cord lies midway between abduction and adduction and a reasonable glottic opening exists. Thus, bilateral incomplete palsy is more dangerous than the complete variety.
TRACHEA
The trachea extends from its attachment to the lower end of the cricoid cartilage, at the level of the 6th cervical vertebra, to its termination at the bronchial bifurcation In the preserved dissecting-room cadaver, this is at the level of the 4th thoracic vertebra and the manubriosternal junction (the angle of Louis), but in the living subject in the erect position, the lower end of the trachea can be seen in oblique radiographs of the chest to extend to the level of the 5th, or in full inspiration the 6th, thoracic vertebra. In the adult, the trachea is 15 cm long, of which 5 cm lie above the suprasternal notch; this portion is somewhat greater (nearly 8 cm) when the neck is fully extended. The diameter of the trachea is correlated with the size of the subject; a good working rule is that it has the same diameter as the patient's index finger. The patency of the trachea is due to a series of 16–20 C-shaped cartilages joined vertically by fibroelastic tissue and closed posteriorly by the nonstriated trachealis muscle. The cartilage at the tracheal bifurcation is the keel-shaped carina, which is seen as a very obvious sagittal ridge when the trachea is inspected bronchoscopically. Should the sharp edge of the carina become flattened, this usually denotes enlargement of the hilar lymph nodes or gross distortion of the pulmonary anatomy by fibrosis, tumor or other pathology
Relations
The trachea lies exactly in the midline in the cervical part of its course but within the thorax it is deviated slightly to the right by the arch of the aorta. In the neck, it is covered anteriorly by the skin and by the superficial and deep fascia, through which the rings are easily felt. The 2nd to the 4th rings are covered by the isthmus of the thyroid where, along the upper border, branches of the superior thyroid artery join from either side. In the lower part of the neck, the edges of the sternohyoid and sternothyroid muscles overlap the trachea, which is here also covered by the inferior thyroid veins 14(as they stream downwards to the brachiocephalic veins), by the cross-communication between the anterior jugular veins and, when present, by the thyroidea ima artery, which ascends from the arch of the aorta or from the brachiocephalic artery. It is because of this close relationship with the brachiocephalic artery that erosion of the tracheal wall by a tracheostomy tube may cause sudden profuse hemorrhage. It is less common for the carotid artery to be involved in this way. On either side are the lateral lobes of the thyroid gland, which intervene between the trachea and the carotid sheath and its contents (the common carotid artery, the internal jugular vein and the vagus nerve). Posteriorly, the trachea rests on the esophagus, with the recurrent laryngeal nerves lying on either side in a groove between the two. The close relationship of the unsupported posterior tracheal wall and the esophagus is revealed during esophagoscopy. The thoracic part of the trachea descends through the superior mediastinum. Anteriorly, from above downwards, lie the inferior thyroid veins, the origins of the sternothyroid muscles from the back of the manubrium, the remains of the thymus, the brachiocephalic artery and the left common carotid arterya which separate the trachea from the left brachiocephalic vein and, lastly, the arch of the aorta. Posteriorly, as in its cervical course, the trachea lies throughout on the esophagus, with the left recurrent laryngeal nerve placed in a groove between the left borders of these two structures. On the right side, the trachea is in contact with the mediastinal pleura, except where it is separated by the azygos vein and the right vagus nerve. On the left, the left common carotid and left subclavian arteries, the aortic arch and the left vagus intervene between the trachea and the pleura; the altering relationships between the major arteries and the trachea are due to the diverging, somewhat spiral, course of the arteries from their aortic origins to the root of the neck. The large tracheobronchial lymph nodes lie at the sides of the trachea and in the angle between the two bronchi. In infants, these relationships are somewhat modified; the brachiocephalic artery is higher and crosses the trachea just as it descends behind the suprasternal notch. The left brachiocephalic vein may project upwards into the neck to form an anterior relation of the cervical trachea frightening encounter, if found tensely distended with blood when performing a tracheotomy on an asphyxiating baby. In children up to the age of 2 years, the thymus is large and lies in front of the lower part of the cervical trachea.
THE MAIN BRONCHUS
The trachea bifurcates in the supine cadaver at the level of the 4th thoracic vertebra into the right and left bronchi. In the erect position in full inspiration in life, the level of bifurcation is at T6. The right main bronchus is shorter, wider and more vertically placed than the left: shorter because it gives off its upper lobe bronchus sooner (after a course of only 2.5 cm); 15wider because it supplies the larger lung; and more vertically placed (at 25° to the vertical compared with 45° on the left) because the left bronchus has to extend laterally behind the aortic arch to reach its lung hilum. Thus, inhaled foreign bodies are more inclined to enter the wider and more vertical right bronchus than the narrower and more obliquely placed left. The right pulmonary artery is first below and then in front of the right main bronchus, and the azygos vein arches over it. The left main bronchus is 5 cm long. It passes under the aortic arch, in front of the esophagus, thoracic duct and descending aorta, and has the left pulmonary artery lying first above and then in front of it. Because the right upper lobe bronchus arises only a short distance below the carina, it is not possible to place a tube in that bronchus without the risk of obstruction of the lower lobe. To overcome this difficulty, right-sided endobronchial tubes have an orifice in the lateral surface of the tube that coincides with the opening of the right upper lobe. No special arrangement has to be made for tubes placed in the left bronchus, as the 5 cm distance between the carina and the left upper lobe bronchus leaves ample room for the cuffed end of an endobronchial tube.
The Lungs (Fig. 13)
Each lung is roughly conical, with an apex, a base, a lateral (or costal) and a medial surface and with three borders—anterior, posterior and inferior. Each lung lies freely within its pleural cavity apart from its attachments at the hilum. The right lung is the larger, weighing on average 620 g compared with 570 g on the left. The lung of the male is larger and heavier than that of the female. Each lung is divided by a deep oblique fissure, and the right lung is further divided by a transverse fissure. Thus, the right lung is trilobed and the left bilobed.
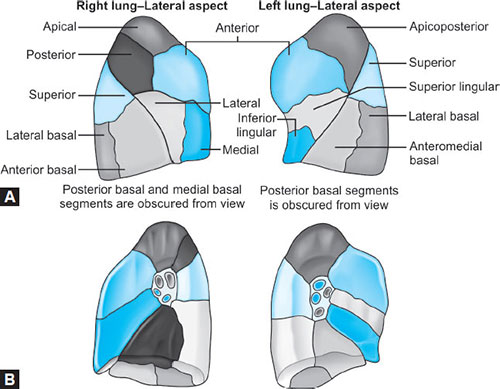
Bronchopulmonary Segments (Figs. 14A and B)
Characteristics | Children (up to 1 year) | Adults |
|---|---|---|
Carina cricoid distance | 5-6 cm | 10-20 cm |
Right bronchus angle | 30 (straighter) | 20 |
Left bronchus angle | 45 | 45 |
Narrowest part of airway | Cricoid | Glottis |
Glottic level | C3-4 interspace | C5 |
Vocal cords | Anteroinferior | Horizontal |
Epiglottis | Omega shaped (long, anterior) | Crescent |
Corniculate/cuneiform tubercles | Prominent | Minimal |
Glottic-epiglottic angle | Small | Large |
Prominence of occiput | Large head (compared to body) | Small |
Breathing route | Nasal | Mouth or nasal |
Laryngoscope preferred | Straight blade | Curved blade |
PHYSIOLOGY
Geetanjali S Verma
FUNCTIONAL ANATOMY
The nose, mouth and pharynx conduct air to the larynx, humidify and filter the air gases. The larynx aids phonation and conducts the gas into the trachea (18 mm diameter and 11 cm length).
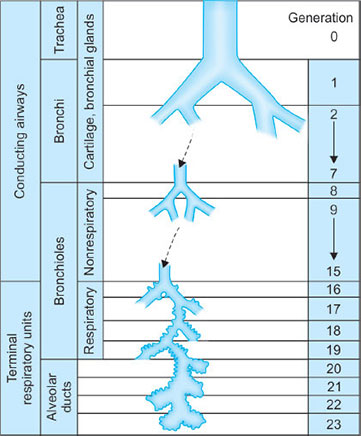
It is lined with columnar ciliated epithelium and divides into the left and right major bronchi at the carina (T4). The bronchi divide 23 times in total (23 generations) (Fig. 16) in order to increase the surface area available for gas exchange. The first 16 generations are termed the conducting zone (no bronchi in his region take part in gas exchange and this forms the anatomical dead space). In an average adult the volume of this space is about 150 mL. From generation 17, small alveoli bud off the bronchi. Generation 17–23 is the respiratory zone where gas exchange occurs. The volume of this zone is about 2–3 liters and there are about 300 million alveoli present within an average lung.
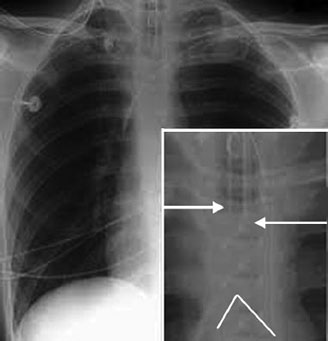
- The endotracheal tube (ETT) in an adult should lie 1–2 cm superior to the carina (Fig. 17)
- On an X-ray the carina is the point at which the trachea can be seen dividing into the right and left bronchi—around T4.
STRUCTURE AND FUNCTION OF RESPIRATORY SYSTEM
- Exchange of O2 and CO2
- Blood reservoir
- Heat exchange
- Metabolism—synthesis and catabolism
- Immunological and mechanical defence blood/gas barrier to diffusion ~50–80 m2 alveolar walls have two sides
- active side ~0.4 mm
- service side ~1–2 mm.
Type | Function | Structure |
|---|---|---|
Conductive | Bulk gas movement | Trachea to terminal bronchioles |
Transitional | Bulk gas movement Limited gas exchange | Respiratory bronchioles Alveolar ducts |
Respiratory | Gas exchange | Alveoli Alveolar sacs |
Cells Types and Functions in the Lung
Type 1 alveolar cells: Derived from type II alveolar cells, provide a thin layer of cytoplasm which covers about 80% of the gas exchange zone. 0.1 mm thick, have 1 nm gap junctions, impermeable to albumin, allow extravasation of mf's—unable to divide—highly sensitive to hyperoxia.
Type II alveolar cells: These cells allow the formation of surfactant and other enzymes. They are rounded cells at septal junctions, resistant to hyperoxia.
Type III alveolar cells: These cells are the main lung defence system—alveolar macrophages.
SURFACE TENSION
A thin film of liquid lines the alveoli and the surface tension of this film is an important factor in the pressure-volume relationship of the lung. The surface tension arises because the attractive forces between adjacent molecules of the liquid are much stronger than those between the liquid and the gas. As a result of that the liquid surface area becomes as small as possible. At the interface between the liquid and the alveolar gas, intermolecular forces in the liquid tend to cause the area of the lining to shrink (the alveoli tend to get smaller). The surface tension contributes to the pressure-volume behavior of the lungs because when the lungs are inflated with saline they have much larger compliance that when they are filled with air (because saline abolishes the surface tension).
This generates a pressure predicted from Laplace's law:
Pressure = (4 × surface tension)/radius
the surface tension contributes a large part of the static recoil force of the lung (expiration).
The surface tension changes with the surface area: The larger the area, the smaller the surface tension.
SURFACTANT
Surfactant is stored in the lamellar bodies of type II alveolar cells and made up of phospholipids, plasma proteins and carbohydrate. It is an amphipathic molecule with a charged hydrophilic head and hydrophobic tail.
Its major constituent is dipalmitoyl phosphatidylcholine (DPPC), which is synthesized in the lung from fatty acids that are either extracted from the blood or are themselves synthesized in the lung.
Functions of surfactant are:
- Reduces surface tension within the alveoli which helps to increase the compliance of the lung
- Improves alveolar stability
- Keeps alveoli dry by opposing water movement from the pulmonary interstitium.
APPLIED PHYSIOLOGY
Lung Volumes and Capacities (Fig. 18)
Primary lung volumes:
- Residual volume (RV)
- Expiratory reserve volume (ERV)
- Tidal volume (TV)
- Inspiratory reserve volume (IRV)
Secondary derived capacities:
- Total lung capacity (TLC)
- Vital capacity (VC)
- Inspiratory capacity (IC)
- Functional residual capacity (FRC)
Lung volumes vary with age, sex, height and weight, and are formulated into normograms.
Definitions
Residual volume (RV) | Volume of gas remaining in lungs after a forced expiration | 15-20 mL/kg |
Expiratory reserve volume (ERV) | Volume of gas forcefully expired after normal tidal expiration | 15 mL/kg |
Tidal volume (TV) | Volume of gas inspired and expired during normal breathing | 6 mL/kg |
Inspiratory reserve volume (IRV) | Volume of gas inspired over normal tidal inspiration | 45 mL/kg |
Total lung capacity | Volume of gas in lungs at the end of maximal inspiration | 80 mL/kg |
Vital capacity | IRV + TV + ERV | 60-70 mL/kg |
FRC | ERV + RV | 30 mL/kg |
(Any 2 or more volumes added together make a capacity) | ||
Functional Residual Capacity
The volume of gas left in the lungs at the end of normal tidal expiration
FRC = ERV + RV
FRC acts as a buffer:
- Maintaining relatively constant A and a gas tensions with each breath
- Preventing rapid changes in alveolar gas with changes in ventilation or inspired gas, e.g. during induction or recovery from anesthesia
- Increasing the average lung volume during quiet breathing, reducing work of breathing due to shape of compliance curve.
Factors decreasing FRC:
Age, posture—supine position, anesthesia—muscle relaxants, surger—laparoscopic, pulmonary fibrosis/pulmonary edema, obesity, abdominal swelling, pregnancy—increased abdominal pressure.
Factors increasing FRC:
Increasing height of patient, erect position—diaphragm and abdominal organs less able to encroach upon bases of the lungs, emphysema—decreased elastic recoil of lung therefore less tendency of lung to collapse, asthma—air trapping.
Measurement
- Helium dilution
- Body plethysmography
- Nitrogen washout.
Closing Capacity (CC)
This is the volume at which the small airways close during expiration. Under normal circumstances the FRC is always greater than the CC however, if the FRC was to decrease then this would no longer be the case and the small airways may close at the end of normal tidal expiration. This leads to hypoxemia, atelectasis and worsening gas exchange due to increasing V/Q mismatch.
Closing capacity increases with age.
Typically closing capacity = FRC at the age of 66 years in the erect position or 44 years in the supine position (Fig. 19).23
Fig. 19: Relation of FRC & CC.Source: www.frca.co.uk/Documents/147%20Respiratory%20physiology%20part%201.pdf
Definition of Respiratory Pattern Terminology
Word | Definition |
|---|---|
Eupnea | “Good breathing”: continuous inspiratory and expiratory movement without interruption |
Apnea | “No breathing”: cessation of ventilatory effort at passive end-expiration (lung volume = FRC) |
Apneusis | Cessation of ventilatory effort with lungs filled at TLC |
Apneustic ventilation | Apneusis with periodic expiratory spasms |
Biot | Ventilatory gasps interposed between periods of ventilation apnea; also “agonal ventilation”. |
VENTILATION
Airways and Airflow
Inhaled air passes through the conducting airways and eventually reaches the respiratory epithelium of the lungs. The trachea divides into right and left main bronchi, which in turn divide into lobar, then segmental 12 bronchi. 24This process continues down to the terminal bronchioles (the smallest airways without alveoli).
Since the conducting airways have no alveoli they do not take part in gas exchange but constitute the anatomical dead space (about 150 mL).
During inspiration, respiratory tubes are lengthened and dilated, especially in deep breathing. Since the airways serve as a barrier as well, harmful foreign material including most microorganisms can not easily enter the lower respiratory passages. The very first barrier starts at the vestibules of the nose, which contain hairs, and healthy, sticky mucus intercepting air-borne particles. Caught particles are then ejected by ciliated epithelium, which covers the entire upper respiratory tract.
The larynx and the bifurcation of the trachea are the most sensitive regions and any particles of foreign matter lodged in these regions are removed with a cough reflex.
The alveolated region of the lung includes respiratory bronchioles (divided from terminal bronchioles and have only occasional alveoli on their walls) and alveolar ducts (completely lined with alveoli). This zone is called respiratory zone and the gas exchange occurs here. The distance from the terminal bronchiole to the distal alveolus is only a few mm, but the respiratory zone makes up most of the lung (2.5–3 L).
Blood is brought to the other side of the blood-gas barrier from the right heart by pulmonary arteries, which also form a series of branching tubes leading to the pulmonary capillaries and back to the pulmonary veins. The capillaries lie in the walls of the alveoli and form a dense network that the blood continuously runs in the alveolar wall. At rest, all the capillaries are not open but when the pressure rises (e.g. exercise) recruitment of the closed capillaries occurs. The diameter of a capillary segment is about 10 micrometer (= size of RBC). The pulmonary artery receives the whole output of the right heart, but resistance of pulmonary circuit is very low. This enables the high blood flow to the circuit.
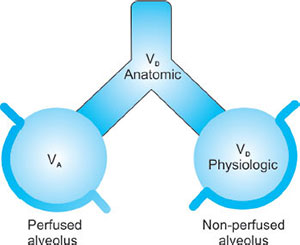
DEAD SPACE
Types
- Anatomical dead space: This includes any breathing system or airway plus mouth, trachea and the airways up until the start of the respiratory zone—does not take part in air exchange.The typical volume in an adult is about 150 mL.
- Alveolar dead space: This occurs when areas of the lung are being ventilated but not being perfused and this leads to what is known as V/Q mismatch.25Large increases in alveolar dead space commonly occur in the following conditions: pneumonia, pulmonary edema, and pulmonary embolism
- Physiological dead space = alveolar + anatomical dead space.Dead space is usually 30% of VT.
Measurement of Dead Space
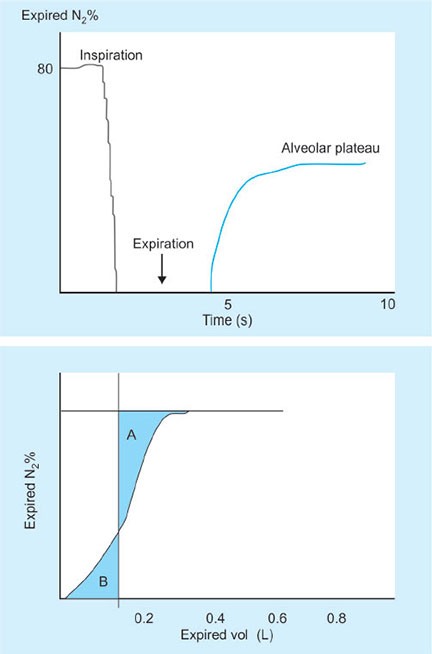
- Fowler's method—tracer washout (Fig. 22)
- Single breath analysis using an indicator gas (N2, CO2, O2, He) to mark the transition between dead space and alveolar gas
- The mid-point of the wash-in (where area A = area B below) measures the transition from conducting airways to the transition from dead space to alveolar gas
- In patients with nonuniform distribution of ventilation, i.e. regions of the lung with different time constants, a slow “wash-in” is seen and the method is inaccurate.
- Bohr's method—conservation of mass
For each tidal volume there will be a proportion of dead space (anatomical) but the amount of gas that is left over should take part in gas exchange.
Abbreviations used in equation:
FACO2- Alveolar CO2
FeCO2- CO2 from mixed expired gases
VT- Tidal volume
VD- Dead space volume (physiological)
Bohr's Equation Derivation
VT. FECO2 = VA. FACO2
But VA = VT - VD
Substituting: VT. FECO2 = (VT - VD). FACO2
VT. FECO2 = VT. FACO2 - VD. FACO2
Rearranging: VD. FACO2 = VT. FACO2 - VT. FECO2
= VT (FACO2 - FECO2)
Hence, VD/VT = (FACO2-FECO2)/FACO2
Or VD/VT = PaCO2 - PECO2/PaCO2
RESPIRATORY MECHANICS
Respiratory Muscles
Inspiratory muscles
- Diaphragm—has the ability to contact 10 cm in forced inspiration
- External intercostals—pull the ribs up and forwards
- Accessory inspiratory muscles—scalene muscles (elevate first 2 ribs) and sternomastoids (raise the sternum)
- Muscles of neck and head (seen in small babies in respiratory distress).
Expiratory muscles
Expiration is usually passive and relies on the elastic recoil of the lungs and the chest wall.
Under anesthesia or extreme exercise, expiration may become active due to the activation of abdominal muscles. Muscles have their use in forced expiration.
- Abdominal wall muscles—rectus abdominis, internal and external oblique
- Internal intercostal muscles—pull ribs down and inwards.
Elastic recoil is usually measured in terms of compliance.
Compliance is defined as the volume change per unit pressure change.
Compliance = ΔV/ΔP
It is classified into chest wall, lung or total lung compliance (distensibility).
Normally, it equals 200 mL/cm H2O
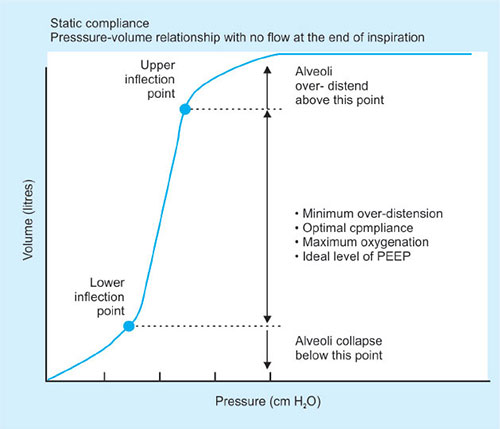
There are 2 types of compliance: static and dynamic
Static compliance is measured during plateau pressure.
Dynamic compliance varies and is calculated with measurement of tidal volume at a given intrathoracic pressure during which there is airflow through the lungs at any point during inspiration or expiration.
A variety of factors affect this like lung volume, pulmonary blood volume, extravascular lung water and pathological processes (inflammation, fibrosis)
Compliance can be measured by inserting an esophageal probe into a cooperative patient, the patient inhales and exhales to a set volume. At each volume the intrapleural pressure is estimated using the esophageal probe. A pressure volume curve can then be plotted. If during the measurement process no gas flow occurs at each set volume then this is static compliance. (Gas flow ceases and equilibration occurs.) If gas flow continues throughout measurement then this is dynamic compliance.
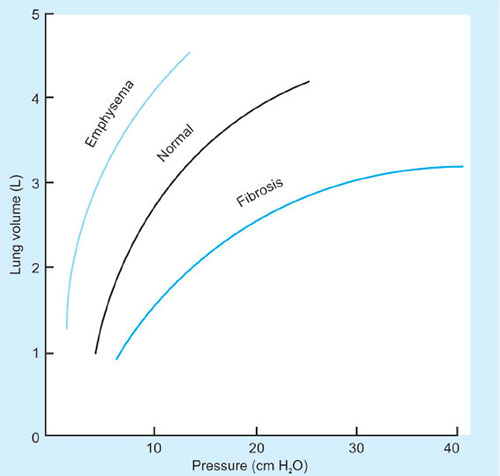
Compliance increases in old age and emphysema as elastic lung tissue is destroyed. It is decreased in pulmonary fibrosis, pulmonary edema, atelectasis and in the extremes of lung volume.
Factors affecting compliance are:
Disease: In atelectasis, when the lung is relatively stiff, the point of balance (resting expiratory volume) will be reached at a lower lung volume, as there will be greater pull inwards; this will predispose to further atelectasis. The excessively compliant emphysematous lung has less elastic recoil; resting expiratory volume will then be greater, since the natural tendency for the chest wall to expand is maintained.
Age: The immature lung in the infant is less elastic than in adulthood. Elasticity is highest in young adults and it decreases slowly with advancing age—lung compliance is therefore lowest in young adults. Chest wall compliance is highest at birth and slowly declines with age.
Posture: Thoracic compliance is lower in the supine position, as the gravitational pull of the abdomen, which existed in the upright position, is reversed and the diaphragm is pushed into the chest by the abdominal contents.
Anesthesia: Several factors (supine position, airway closure, changes in intrathoracic blood volume, accumulation of fluid, direct effect of drugs, altered muscle tone, external pressure) influence compliance under anesthesia; generally compliance is decreased.
Obesity: The effect is compounded by supine or lithotomy position.
Work of Breathing
It is the work required by the respiratory muscles to overcome the mechanical impedance to respiration. It is the sum of work requires to overcome both elastic and airflow resistance.
The energy required for the work of breathing is mainly used in the process of inspiration as energy is required to overcome airway resistance, the elastic recoil of the tissues and the chest wall and tissue resistance. The energy stored within the elastic tissues is used to provide for expiration.
- With the work to overcome elastic resistance given by the trapezoid OAECGO the difference between these representing the nonelastic resistance, given by the area ABCEA.
- This is not the work of “breathing”, as some work is performed by the stored elastic potential energy of the thoracic cage.
- The true work of inspiration is given by ABCDA, with the elastic component being AECDA.
- As airway resistance, or inspiratory flow rate is increased, so would dPIP, effectively sloping the curve to right, increasing total and viscous work on expiration.
- The work to overcome nonelastic forces (AECFA), falls within work trapezoid and can be accomplished with the stored energy in elastic structures the difference between AECDA-AECFA represents the energy expenditure with which no external work is done is released as heat.
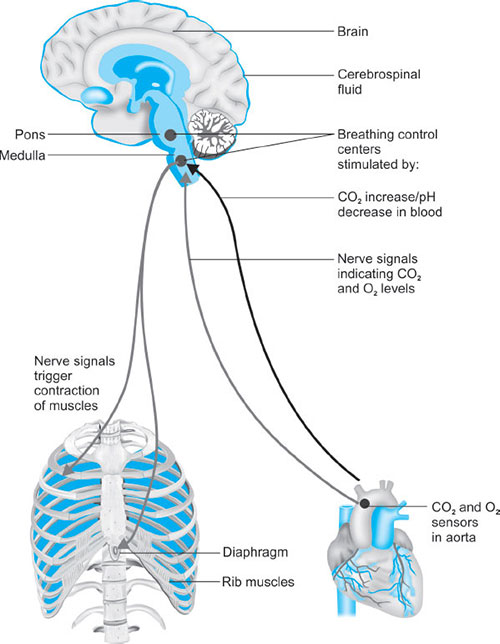
REGULATION AND CONTROL OF BREATHING (FIG. 26)
Central Control
Breathing is mainly controled at the level of brainstem. The normal automatic and periodic nature of breathing is triggered and controled by the respiratory centers located in the pons and medulla.
Medullary Respiratory Center
- Dorsal medullary respiratory neurones: Associated with inspiration. It has been proposed that spontaneous intrinsic periodic firing of these neurones responsible for the basic rhythm of breathing.32As a result, these neurones exhibit a cycle of activity that arises spontaneously every few seconds and establish the basic rhythm of the respiration. When the neurones are active their action potentials travel through reticulospinal tract in the spinal cord and phrenic and intercostal nerves and finally stimulate the respiratory muscles.
- Ventral medullary respiratory neurones: Associated with control of breathing. These neurones are silent during quite breathing because expiration is a passive event following an active inspiration. However, they are activated during forced expiration when the rate and the depth 33of the respiration is increased (e.g. exercise). During heavy breathing increased activity of the inspiratory center neurones activates the expiratory system. In turn, the increased activity of the expiratory system inhibits the inspiratory center and stimulates muscles of expiration.
The dorsal and ventral groups are bilaterally paired and there is 8 cross communication between them. As a consequence they behave in synchrony and the respiratory movements are symmetric.
Apneustic Center
It is located in the lower pons.
Exact role is not known. Lesions covering this area in the pons cause a pathologic respiratory rhythm with increased apnea frequency. What is known is nerve impulses from the apneustic center stimulate the inspiratory center and without constant influence of this center respiration becomes shallow and irregular.
Pneumotaxic Center
It is located in the upper pons.
These neurones have an inhibitory effect on the both inspiratory and apneustic centers. It is probably responsible for the termination of inspiration by inhibiting the activity of the dorsal medullar neurones. It primarily regulates the volume and secondarily the rate of the respiration. In the lesions of this area normal respiration is protected, thus it is believed that upper pons is responsible for the fine-tuning of the respiratory rhythm. Hypoactivation of this center causes prolonged deep inspirations and brief, limited expirations by allowing the inspiration center remain active longer than normal. Hyperactivation of this center on the other hand results in shallow inspirations.
The apneustic and pneumotaxic centers function in coordination in order to provide a rhythmic respiratory cycle: Activation of the inspiratory center stimulates the muscles of inspiration and also the pneumotaxic center. Then the pneumotaxic center inhibits both the apneustic and the inspiratory centers resulting in initiation of expiration. Spontaneous activity of the neurones in the inspiratory center starts another similar cycle again. Breathing in some extent is also controled consciously from higher brain centers (e.g. cerebral cortex). This control is required when we talk, cough and vomit. It is also possible voluntarily change the rate of the breathing. Hyperventilation can decrease blood partial carbon dioxide pressure (PCO2) due to loss of CO2 resulting in peripheral vasodilatation and decrease in blood pressure. One can also stop breathing voluntarily. That results in an 34increase in arterial partial oxygen pressure (PO2), which produces an urge to breathe. When eventually PCO2 reaches the high enough level it overrides the conscious influences from the cortex and stimulates the inspiratory system. If one holds his breath long enough to decrease PO2 to a very low level one may loose his consciousness. In an unconscious person, automatic control of the respiration takes over and the normal breathing resumes. Other parts of the brain (limbic system, hypothalamus) can also alter the breathing pattern, e.g. affective states, strong emotions, such as rage and fear. In addition, stimulation of touch, thermal and pain receptors can also stimulate the respiratory system.
SENSORS/OTHER RECEPTORS (FIG. 27)
Mechanoreceptors
These receptors are placed in the walls of bronchi and bronchioles of the lung. The main function of these receptors is to prevent the overinflation of the lungs. Inflation of the lungs activates these receptors and activation of the stretch receptors in turn inhibits the neurones in inspiratory center via vagus nerve. When the expiration starts activation of the stretch receptors gradually ceases allowing neurones in the inspiratory neurones become active again (Hering-Breuer Reflex). It is particularly important for infants. In adults it is functional only during exercise when the tidal volume is larger than normal.
Chemoreceptors
The respiratory system maintains concentrations of O2, CO2 and the pH of the body fluids within a normal range. Any deviation from these values has a marked influence on the respiration. Chemoreceptors are specialized neurones activated by changes in O2 or CO2 levels in the blood and the brain tissue, respectively. They are involved in the regulation of respiration according to the changes in PO2 and pH. O2-sensitive chemoreceptors (Peripheral chemoreceptors) are located at the bifurcation of the carotid artery in the neck and the aortic arch. They are small vascular sensory organs encapsulated with the connective tissue. They are connected to the respiratory center in the medulla by glossopharyngeal nerve (carotid body chemoreceptors) and the vagus nerve (aortic body). Central chemoreceptors are located bilaterally in the chemosensitive area of the medulla oblongata and exposed to the cerebrospinal fluid (CSF), local blood flow and local metabolism. They actually respond to changes in H+ concentration in these compartments. When the blood partial PCO2 is increased CO2 diffuses into the CSF from cerebral vessels and liberates H+. (When CO2 combines with water forms carbonic acid and liberates H+ and HCO3−).
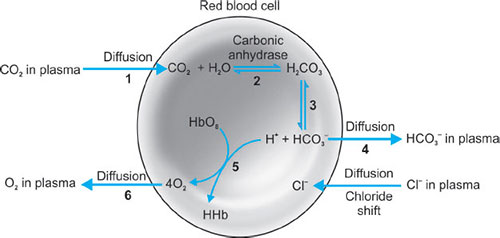
CO2 + H2O↔ H2CO3 H2CO3 ↔ HCO - 3 + H+
An increase in H+ stimulates chemo receptors resulting in hyperventilation which in turn reduces PCO2 in the blood and therefore in the CSF. Cerebral vasodilatation always accompanies an increased PCO2 and enhances the diffusion of CO2 into the CSF. Because CSF has less protein than blood it has a much lower buffering capacity. As a result changes in pH for a given change in PCO2 is always bigger than the change in blood.
CO2 level is a major regulator of respiration. It is much more important than oxygen to maintain normal respiration. Even very small changes in carbon dioxide levels (5 mm Hg increase in PCO2, hypercapnia) in the blood cause large increases in the rate and depth of respiration (100% increase in ventilation). Hypocapnia, lower than normal PCO2 level in the blood causes in periods in which respiratory movements do not occur. Effects of PO2 (if the changes occur within the normal range) on respiration is very minor. A decrease in PO2 is called hypoxia and only after 50% decrease in PO2 can produce significant changes in respiration. This is due to the nature of O2-Hb saturation that at any PO2 level above 80 mm Hg Hb is saturated with O2. Consequently only big changes in PO2 produce symptoms otherwise it is compensated by O2, which is bound with Hb. In stroke patients or physiologically at high altitude blood PO2 level may drop considerably and activate peripheral chemoreceptors and activate stimulation.36
At high altitude because the ability of the lung to eliminate CO2 is not affected, in response to increased respiration, blood PCO2 is decreased. If PO2 drops under certain level respiratory system does not respond and death will occur.
Distribution of Ventilation
Alveolar pressure is the same throughout the lung; therefore, the more negative intrapleural pressure at the apex (or the least gravity-dependent area) results in larger, more distended apical alveoli than in other areas of the lung (Fig. 28). The transpulmonary pressure (Paw – Ppl), or distending pressure of the lung, is greater at the top and lower at the bottom, where intrapleural pressure is less negative. Despite the smaller alveolar size, more ventilation is delivered to dependent pulmonary areas. The decrease in intrapleural pressure at the base of the lungs during inspiration is greater than at the apex because of diaphragmatic proximity. Thus, because the dependent area of the lung generates the greatest change in transpulmonary pressure, more gas is sucked into dependent areas of the lung.
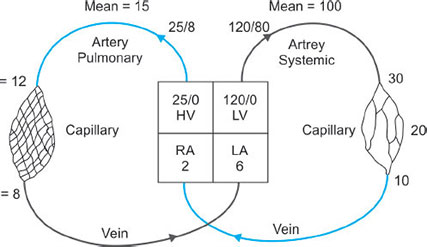
PERFUSION
Blood flow to and around the lung is similar to any other organ but at much lower pressures than the systemic system (Fig. 29).37
The blood vessels in the lungs continually branch and get consistently smaller very like the branching of the airways. The pulmonary arteries whose walls are very thin in comparison to that of the arteries in the main circulation feed the lung up to the level of the terminal bronchioles and then split into the capillary bed. The capillaries have great capability to distend thus enhancing gas exchange and reservoir action. Once the red blood cells have become oxygenated the capillary bed is drained into venules which then join to form the pulmonary veins. It is the ability of the blood vessels to distend and be recruited which allows the pressures in the pulmonary system to stay low despite very high blood flow.
The pulmonary arteries only supply blood flow and oxygen to the lungs and must have the ability to accept huge blood volumes at times. The low pulmonary pressures are important to minimize the work of the right heart.
Pulmonary Vascular Resistance
PVR = 80 × (MPAP – PCWP)/CO
Distribution of Blood Flow
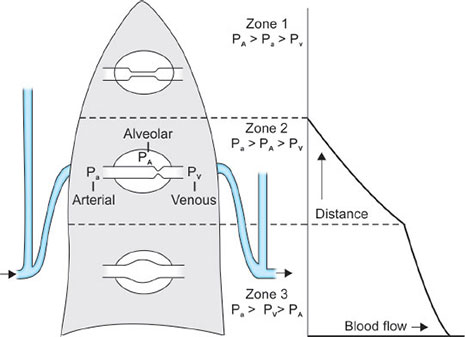
Blood flow within the lung is mainly gravity-dependent. Since the alveolar-capillary beds are not composed of rigid vessels, the pressure of the surrounding tissues can influence the resistance to flow through the individual capillaries. Thus, blood flow depends on the relationship between pulmonary artery pressure (Ppa), alveolar pressure (PA), and pulmonary venous pressure. West et al. and West and Dollery created a lung model which divides the lung into three zones (Fig. 30).
Zone 1 conditions occur in the most gravity-independent part of the lung (alveolar pressure is approximately equal to atmospheric pressure; and pulmonary artery pressure).
Zone 2 occurs from the lower limit of zone 1 to the upper limit of zone 3 = Ppa > Pα > Ppv. The pressure difference between pulmonary artery and alveolar pressure determines blood flow in zone 2. Pulmonary venous pressure has little influence. Well-matched ventilation and perfusion occur in zone 2, which contains the majority of alveoli (Figs. 30 and 31).
Zone 3 occurs in the most gravity-dependent areas of the lung = Ppa > Ppv > PA. blood flow is primarily governed by the pulmonary arterial to venous pressure difference. Because gravity also increases pulmonary venous pressure, the pulmonary capillaries become distended. Thus, perfusion in zone 3 is lush, resulting in capillary perfusion in excess of ventilation, or physiologic shunt.
Schematic representation of the effects of gravity on the distribution of pulmonary blood flow in the lateral decubitus position. Vertical gradients in the lateral decubitus position are similar to those in the upright position and cause the creation of West zones 1, 2, and 3. Consequently, pulmonary blood flow increases with lung dependency, and is largest in the dependent lung and least in the nondependent lung. Pa, pulmonary artery pressure; PA, alveolar pressure; Pv, pulmonary venous pressure.
Distribution of Ventilation and Perfusion
The efficiency with which oxygen and carbon dioxide exchange at the alveolar-capillary level highly depends on the matching of capillary perfusion and alveolar ventilation. At this level, the combination of lung and the circulatory system must be well-matched.
Ventilation-Perfusion Relationships
The majority of blood flow is distributed to the gravity-dependent part of the lung. During a spontaneous breath, the largest portion of the tidal volume also reaches the gravity-dependent part of the lung.40
Thus, the nondependent area of the lung receives a lower proportion of both ventilation and perfusion, and dependent lung receives greater proportions of ventilation and perfusion. But, ventilation and perfusion are not matched perfectly, and various V/Q ratios result throughout the lung (Fig. 32).
Any discrepancy between ventilation and blood flow in the lung will result in V/Q mismatch and potentially dangerous irregularities in gas exchange.
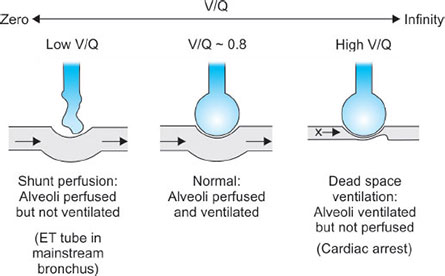
If flow of blood to the lung units is to match that of ventilation to the same unit then the ratio of ventilation to perfusion should be in a ratio of 1:1.
If the lung is being underventilated but perfused as normal then we say that the V/Q ratio is <1 (Fig. 33).
If the lung is under perfused then the V/Q is >1 (Fig. 33).
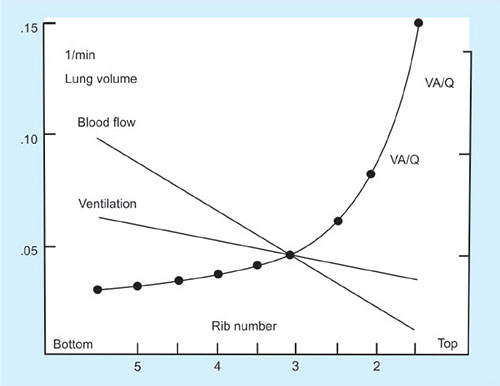
Even in a normal lung the V/Q ratio is not uniformly 1 throughout the lung as perfusion and ventilation both have favored parts of the lung. Differences between the apices and bases of the lungs.
At the apices there is less ventilation than the bases as alveoli are already very stretched however there is proportionally less perfusion therefore the overall V/Q ratio is higher compared to the base of the lung.
Blood flow is directly affected by gravity and naturally has a tendency to flow to the bases of the lungs thus V/Q ratios toward the lower segments of the lung are usually greater than 1. The vertical change in V/Q ratios in the lung is because although both ventilation and perfusion increase from top to bottom of the lung, perfusion increases much quicker than ventilation.41
Thus the V/Q ratio at the top of the lung is 3.3 whereas at the bases it is around 0.6.
Ideal V/Q ratio = 1; believed to occur at approximately the level of the third rib.
Above this level, ventilation occurs slightly in excess of perfusion, whereas below the third rib the V/Q ratio becomes less than 1.
V/Q = 0 in shunt
V/Q = infinity in dead space.
Hypoxic pulmonary vasoconstriction and bronchoconstriction allow the lungs to maintain optimal V/Q matching.
Many pulmonary diseases result in both physiologic shunt and dead space abnormalities. However, most disease processes can be characterized as producing either primarily shunt or dead space in their early stages. Increases in dead space ventilation primarily affect carbon dioxide elimination and have little influence on arterial oxygenation until dead space ventilation exceeds 80 to 90% of minute ventilation. Similarly, physiologic shunt primarily affects arterial oxygenation with little effect on carbon dioxide elimination until the physiologic shunt fraction exceeds 75 to 80% of the cardiac output. Defective to absent gas exchange can be the net effect of either abnormality in the extreme.
GAS EXCHANGE
The partial pressure of oxygen that is inhaled from our natural environment through normal inhalation is not maintained at the same partial pressure by the time it reaches the alveoli and indeed the mitochondria. The process by which this decrease in partial pressure occurs is called the oxygen cascade.42
- Dry atmospheric air gas – 21% of 100 kPa
SO: 21 kPa or 160 mm Hg
- However as gas is inspired it is diluted by water vapor which reduces the partial pressure of oxygen water vapor – 6.3 kPa/47 mm Hg
PO2 = 0.21 × (760 – 47) = 149 mm Hg or
PO2 = 0.21 × (100 – 6.3) = 19.8 kPa
- When the gas reaches the alveoli the partial pressure of oxygen will again decrease as some oxygen is absorbed and CO2 is excreted. The partial pressure at this point in the oxygen cascade can be determined by using the alveolar gas equation.
PAO2 = PIO2 – PACO2/RQ
The RQ stands for respiratory quotient and is normally 0.8.
It is determined by the amount of CO2 produced/oxygen consumed.
PAO2 = 0.21–5/0.8 = 14 kPa (106 mm Hg)
- Again when the gas reaches the arterial blood a further small drop in partial pressure will have occurred as blood known as venous admixture with a lower oxygen content mixes with the oxygenated alveolar blood. Venous admixture is made up of blood that has passed through poorly ventilated regions of lung and thus has a lower O2 partial pressure. Venous admixture is also composed of venous blood which has drained the lungs and left side of the heart. This blood is known as true shunt and drains directly into the left side of the heart. Extraction of oxygen from this blood then causes the end capillary oxygen partial pressure to be 6–7 kPa (40–50 mm Hg)
- In the mitochondria the PO2 varies hugely from 1–5 kPa (7.5–40 mm Hg). This provides us with an explanation for the following graph, the oxygen cascade (Fig. 34).
The speed and ease of diffusion are controlled by the laws of diffusion. Fick's law of diffusion states that gas transfer across a membrane is directly proportional to the concentration gradient.
Graham's law states that diffusion of a gas is inversely proportional to the square root of the molecular weight of the molecule.
Other factors which increase diffusion:
- Large surface area
- Thin membrane
- High solubility
The following equation incorporates the important factors Diffusion is proportional to A/T D (P1–P2):
A = Area T = Thickness D = Diffusion constant P1–P2 = Concentration gradient Diffusion in the lungs can be limited in the presence of disease states, e.g. pulmonary edema and thickening of the alveolar membrane in pulmonary fibrosis.
Oxygen Transport
Oxygen is carried in 2 forms in the blood:
- Oxygen combined to hemoglobin (97%). Hemoglobin molecule consists of 2 alpha and 2 beta chains; each chain is formed from an iron–porphyrin molecule—hem. Each hemoglobin molecule can bind 4 oxygen molecules (20 mL oxygen per 100 mL blood) or 15 mL oxygen per 100 mL in venous blood.
- Oxygen dissolved in the blood—this accounts for a minimal amount (0.3 mL per dL).
The amount dissolved obeys Henrys’ law—amount is proportional to the partial pressure 0.023 mL per kPa per 100 mL blood oxygen content in the blood. Total content of oxygen in the blood can be calculated from the Oxygen flux equation:
Flux = [CO × Hb × Saturation × Huffners constant (1.39)] + (0.023 × PO2)
Oxygen dissociation curve (Fig. 35):
- Sigmoid shaped curve relating the fact that binding of oxygen to the hemoglobin molecule is a cooperative process
- Describes the relationship of saturation of hemoglobin with oxygen at varying partial pressures
- Be aware of the P50 –(point at which Hb is 50% saturated)
- Decreasing pH, increasing temperature, 2,3-DPG and CO2 tension will cause a right shift of the curve
- If a right shift occurs the Hb molecule is more likely to offload oxygen to the tissues
- In a left shifted situation the Hb is less likely to release oxygen to the tissues.
2,3-DPG: This molecule binds to deoxygenated Hb – it reduces the affinity of hemoglobin for oxygen and therefore ensures offloading of oxygen to the tissues.
The Bohr effect: This describes the affect that CO2 has on influencing the release of oxygen to the tissues. On entering red blood cells the following reaction occurs:
CO2 + H2O ↔ H2CO3 ↔ H+ + HCO3−
An increase in H+ will cause an acidosis and therefore encourage the release of oxygen from Hb.
In the lungs where the CO2 is being removed, the alkalosis will encourage the uptake of oxygen.
Carbon Dioxide Transport
Carbon dioxide is carried in the blood in 3 ways:
- As bicarbonate – 90%
- As dissolved CO2 – 5%
- As carbamino compounds – 5%
Carbamino compounds are formed by the reaction of the CO2 with terminal amino groups of proteins and side chains of arginine and lysine. Hemoglobin is essential for this process to occur since it has 4 amino groups per molecule. Albumin also provides amino groups but only 1 per molecule.
The Hamburger effect (chloride shift) (Fig. 36).
The transport of chloride ions into the cell as a result of outwards diffusion of bicarbonate in order to maintain electrical neutrality.
The Haldane effect:
This phenomenon refers to the increased ability of blood to carry CO2 when hemoglobin is deoxygenated. Deoxyhemoglobin is 3.5 times more effective than oxyhemoglobin in forming carbamino compounds.
ALVEOLAR-ARTERIAL PO2 GRADIENT
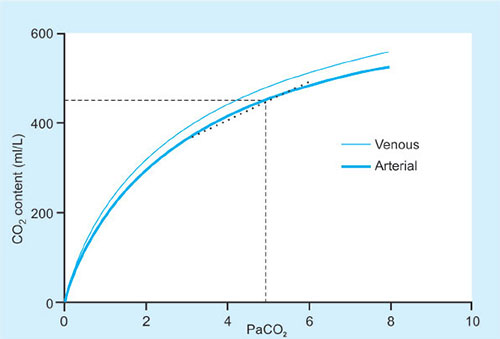
CO2 Dissociation Curve (Fig. 37)
The value for the A-a gradient gives the clinician some idea about the amount of VQ mismatch and shunt that is present in the lungs. A typical normal value would be around 0.5-1 kPa (5 mm Hg) though values up to around 15 mm Hg may be accepted.
- It is calculated as PAO2 – PaO2.
Shunt
True shunt refers to a VQ = 0.
That is to say that blood has passed through areas of the lung where no ventilation is occurring. As discussed earlier VQ mismatch is also referred to as shunt. Blood passes through areas of the lung which are poorly ventilated, i.e. VQ < 1.
Physiological shunt refers to the amount of venous admixture which is directly added to main circulatory blood without having passed through the oxygenating mechanism of the lung. Blood from the bronchial veins draining the lung parenchyma and the thebesian veins draining the cardiac muscle represent the physiological shunt (around 5% of cardiac output). The shunt equation allows calculation of the amount of shunt present in an individual subject.
The shunt equation: Qs = Shunted blood flow Qt = Cardiac output Qt-Qs = Blood flow through the lungs minus the shunted blood CcO2 = Oxygen content of end pulmonary capillary blood CaO2 = Oxygen content of arterial blood CvO2 = Oxygen content of mixed venous blood shunt equation (Fig. 38).
The amount of oxygen leaving the lungs is Qt × CaO2.
This is equal to the shunted blood flow plus the oxygen content from the lung which would be (Qs × CvO2) + (Qt-Qs) × CcO2. (shunt flow × mixed venous O2 content + pulmonary capillary flow × pulmonary capillary O2 content).47
Qt × CaO2 = (Qs × CvO2) + (Qt-Qs) × CcO2
When these equations as rearranged it provides the classic shunt equation:
Qs/Qt = CcO2 – CaO2/ CcO2 – CvO2
Carbon Monoxide Diffusing Capacity
Because PO2 in the pulmonary capillary blood varies with time as it moves through the pulmonary capillary bed, oxygen cannot be used to assess diffusing capacity. A gas mixture containing carbon monoxide is the traditional diagnostic gas used to measure diffusing capacity. Its partial pressure in the blood is nearly zero, and its affinity for hemoglobin is 200 times that of oxygen. Carbon monoxide diffusing capacity (DLCO) collectively measures all the factors that affect the diffusion of gas across the alveolar-capillary membrane. The DLCO is recorded in mL CO/min/mm Hg at standard temperature and pressure, dry.
In persons with normal hemoglobin concentrations and normal V/Q matching, the main factor limiting diffusion is the alveolar-capillary membrane. Small amounts of carbon dioxide and inspired gas can produce measurable changes in the concentration of inspired gas compared with expired gas. There are several methods for determining DLCO, but all methods measure diffusing capacity according to the equation:
The average value for resting subjects when the single-breath method is used is 25 mL CO/min/mm Hg. DLCO values can increase to 2 or 3 times normal during exercise.48
The DLO2 may be estimated from the DLCO by multiplying DLCO by 1.23, although the DLCO is usually the reported value. DLCO can be divided by the lung volume at which the measurement was made to obtain an expression of diffusing capacity per unit lung volume.
Some of the other factors that can influence DLCO are as follows:
- Hemoglobin concentration: Decreased hemoglobin concentration decreases the DLCO.
- Alveolar PCO2: An increased PACO2 raises DLCO.
- Body position: The supine position increases DLCO.
- Pulmonary capillary blood volume.
- Diffusing capacity is decreased in alveolar fibrosis associated with sarcoidosis, asbestosis, berylliosis, oxygen toxicity, and pulmonary edema. These states are frequently categorized as diffusion defects, but low DLCO is probably more closely related to loss of lung volume or capillary bed perfusion.
DLCO is decreased in obstructive disease because of the decreased alveolar surface area, loss of capillary bed, the increased distance from the terminal bronchiole to the alveolar-capillary membrane, and V/Q mismatching. In short, few disease states truly inhibit oxygen diffusion across the alveolar-capillary membrane.
CLINICAL APPLICATIONS
Pulmonary Function Testing
Used in patients with significant pulmonary dysfunction.
Pulmonary Function Tests in Restrictive and Obstructive Lung Disease.
Value | Restrictive disease | Obstructive disease |
|---|---|---|
Definition | Proportional decreases in all lung volumes | Small airway obstruction to expiratory flow |
FVC | ↓↓↓ | Normal or slightly ↑ |
FEV1 | ↓↓↓ | Normal or slightly ↓ |
FEV1/FVC | Normal | ↓↓↓ |
FEF25 −75% | Normal | ↓↓↓ |
FRC | ↓↓↓ | Normal or ↑, if gas trapping |
TLC | ↓↓↓ | Normal or ↑, if gas trapping |
Flow-Volume Loops (Fig. 39)
The flow-volume loop graphically demonstrates the flow generated during a forced expiratory maneuver followed by a forced inspiratory maneuver, plotted against the volume of gas expired.49
The subject forcefully exhales completely, then immediately and forcefully inhales to vital capacity. The expired and inspired volumes are plotted on the abscissa and flow is plotted on the ordinate. Although various numbers can be generated from the flow-volume loop, the configuration of the loop itself is probably the most informative part of the test. It helps in distinguishing an extrathoracic from intra thoracic obstruction and guide in its management.50
Respiratory Formulas
Formula | Normal values (70 kg) |
|---|---|
Alveolar oxygen tension | 110 mm Hg |
PAO2 = (PB − 47) FIO2-; (PACO2/R) | (FIO2 = 0.21) |
Alveolar-arterial oxygen gradient | <10 mm Hg |
(A - aO2) = PAO2 - PaO2 | (FIO2 = 0.21) |
Arterial-to-alveolar oxygen ratio, PaO2/PAO2 ratio | >0.75 |
Arterial oxygen content | 20 mL/100 mL blood |
CaO2 = (SaO2) (Hb × 1.34) + PaO2 (0.0031) | |
Mixed venous oxygen content | 15 mL/100 mL blood |
C[v with bar above]O2 = (S [v with bar above] O2) (Hb × 1.34) + P[v with bar above]O2 (0.0031) | |
Arterial-venous oxygen content difference | 4-6 mL/100 mL blood |
C(a-[v with bar above])O2 = CaO2 - C[v with bar above]O2 | |
Intrapulmonary shunt | <5% |
[Q with dot above]sp/[Q with dot above]T = (Cc'O2 - CaO2)/(Cc'O2 - C[v with bar above]O2) where Cc'O2 = (Hb × 1.34) + (PAO2 × 0.0031) | |
Physiologic dead space | 0.33 |
VD/VT = (PaCO2 - P  | |
Oxygen consumption | 250 mL/min |
VO2 = CO (CaO2 - CVO2) | |
Oxygen transport | 1,000 mL/min |
DO2 = CO (CaO2) | |
Respiratory quotient | 0.8 |
[V with dot above]CO2/[V with dot above]O2 = R | |
(PAO2: Alveolar oxygen tension; PB: Barometric pressure; FIO2: Fraction inspired oxygen; PACO2: Alveolar carbon dioxide tension; R: Respiratory quotient; PaO2: Arterial oxygen tension; CaO2: Arterial oxygen content; SaO2: Arterial oxygen saturation; Hb: Hemoglobin concentration; Cv-O2: Mixed venous oxygen content; Sv-O2: Mixed venous oxygen saturation; Pv-O2: Mixed venous oxygen tension; [Q with dot above]SP/[Q with dot above]T: intrapulmonary shunt; Cc'O2: End-pulmonary capillary oxygen content; VD: Dead space gas volume; VT: Tidal volume; PaCO2: Arterial carbon dioxide tension; P  | |
RESPONSES TO AIRWAY MANIPULATION
Geetanjali S Verma
Direct laryngoscopy and introduction of tracheal tube are noxious stimuli that can provoke a response of cardiovascular, respiratory and other physiological systems.
RESPONSES (FIG. 40)
Cardiovascular System
Hypertension, tachycardia—exaggerated with increased duration an force of laryngoscopy. Increase in arterial pressures starts within 5 seconds of laryngoscopy, peaks in 1–2 minutes and returns to control levels in 5 minutes. Also noted with use of ILMA.
Respiratory System
Laryngospasm—if laryngoscopy attempted during light plane of anesthesia; bronchospasm, coughing, bucking.
Central Nervous System
Elevation of ICT/IOP.
Fig. 40: Response to tracheal intubation.Source: Bedford RF: Circulatory responses to tracheal intubation. Probl Anesth.
Others
Vomiting, aspiration, latex allergy (Patients with spina bifida, rubber industry workers, atopic patients, and patients with a multiple surgery history are most at risk. Patients with type I hypersensitivity are at risk for developing anaphylaxis with hypotension, rash, and bronchospasm).
PATHWAYS/MECHANISMS
- Stimulation of proprioceptors (especially mechanoreceptors) located in supraglottic and tracheal region (consist of small-diameter myelinated fibers, slowly-adapting stretch receptors with large-diameter myelinated fibers, and polymodal endings of nonmyelinated nerve fibers) cause laryngospasm, tachycardia and hypertension. Laryngospasm and bradycardia is common in children.
- Stimulation of glossopharyngeal and vagus nerves—causes widespread autonomic activation of the sympathetic and parasympathetic nervous systems, causing bradycardia and laryngospasm.
- Stimulation of cardioaccelerator nerves and sympathetic nervous system - causing hypertension and tachycardia (more common in adults) due to release of norepinephrine from adrenergic nerve terminals and secretion of epinephrine from the adrenal medulla.
- Activation of the renin-angiotensin system (including release of renin from the renal juxtaglomerular apparatus, which is innervated by β-adrenergic nerve terminals) causing hypertension.
- Stimulation of CNS (especially in patients with pre-existing neuropathology) - elevation of ICT - hypertension and bradycardia (cushing response).
- Reduced cardiac output - due to PEEP after intubation.An increase in mean intrathoracic pressure due to positive-pressure ventilation (PPV) is transmitted to the thin-walled, compressible superior and inferior venae cave, elevating the downstream pressure for venous return and thereby reducing venous blood return to the right atrium. Because the left side of the heart can only pump what the right side delivers, cardiac output and subsequently arterial BP may fall with PPV.
- Adverse effects of drugs:
- Succinylcholine—bradycardia, fasciculations
- Atracurium, mivacurium—histamine release—tachycardia
- Pancuronium—tachycardia
All the above responses are exaggerated inpatients with pre-existing cardiovascular compromise or intravascular volume depletion.53
The neuroendocrine responses to airway manipulation resulting in tachycardia and HTN may result in a variety of complications in patients with cardiac disease, especially myocardial ischemia—seen as ischemic electrocardiographic ST-segment depression and increased pulmonary artery diastolic blood pressure (BP) during intubation in patients with arteriosclerosis.
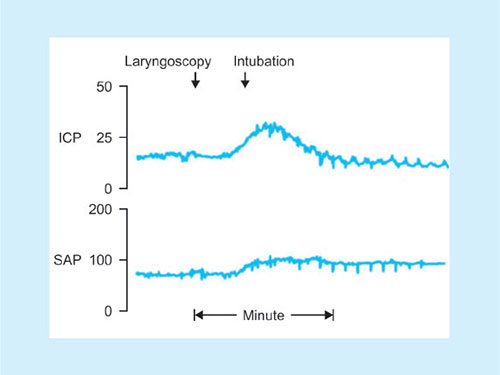
Increases in systemic arterial pressure (SAP) and intracranial pressure (ICP) in response to endotracheal intubation in a patient with a small brain tumor. Notice the minimal response to rigid laryngoscopy. There is a sustained increase in systemic arterial pressure but only a transient increase in ICP, which returns to normal as cerebrovascular autoregulation becomes operative.
PREVENTION OF RESPONSES
- Limit laryngoscopy to <15 seconds, minimize attempts.
- Limit use of cricoid pressure.
- Use of LMA wherever possible.
- Use of topical and regional anesthesia.
- Drugs—fentanyl, propofol, thiopentone, vecuronium (3 minutes preoxygenation).Use of narcotics—fentanyl 6 μg/kg suppress the response, but can cause respiratory depression.
- Xylocard—1–1.5 mg/kg IV 90 seconds prior to intubation.
- Other drugs: Pretreatment with phentolamine, 5 mg IV, prevented the hypertensive-tachycardic response to endotracheal intubation during a light barbiturate-succinylcholine anesthetic technique.Others: Diltiazem, verapamil, and nicardipine, hydralazine, nitroprusside; nitroglycerin; labetalol esmolol; and clonidine.
- Inhalational agents - sevoflurane (MAC > 2.5) blunts hemodynamic responses to intubation.
- Use of N2O has proved beneficial.
- Use of awake flexible fiberoptic intubation with effective topical anesthesia eliminates hemodynamic responses to intubation.
- Use of bronchodilator therapy (in asthmatics) has shown to decrease bronchospasm incidence.